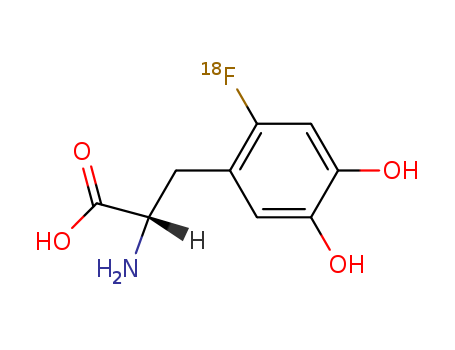
- Chemical Name:Fluorodopa (18F)
- CAS No.:92812-82-3
- Deprecated CAS:167569-09-7,97097-51-3,191087-23-7
- Molecular Formula:C9H10 F N O4
- Molecular Weight:214.183
- Hs Code.:
- UNII:2C598205QX
- DSSTox Substance ID:DTXSID60239157
- Wikidata:Q27121509
- NCI Thesaurus Code:C95766
- RXCUI:2571853
- Metabolomics Workbench ID:154626
- ChEMBL ID:CHEMBL3400972
- Mol file:92812-82-3.mol
Synonyms:(18)F-dopa;18F-FDOPA;2-fluoro-5-hydroxytyrosine;3,4-dihydroxy-6-fluorophenylalanine;3-(2-fluoro-(18)F-4,5-dihydroxyphenyl)-L-alanine;6-(18F)fluoro-L-3,4-dihydroxyphenylalanine;6-(18F)fluoro-L-DOPA;6-fluoro-DOPA;6-fluorodopa;fluorodopa;fluorodopa F 18;fluorodopa F 18, (18)F-labeled cpd;fluorodopa F-18