10.1016/j.tet.2007.08.056
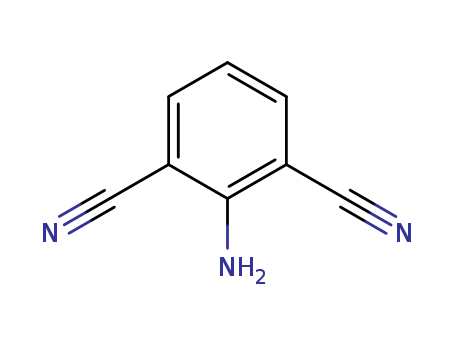
The research describes an efficient and convenient procedure for synthesizing highly functionalized 2-amino-isophthalonitriles and their biaryl compounds. The purpose of this study was to develop a simple, general, and efficient route for the synthesis of these compounds, which possess important biological properties and are useful precursors for the synthesis of other compounds like quinazolines and fluorenones. 2-amino-isophthalonitriles are described as highly functionalized benzene derivatives that possess significant biological properties and serve as useful precursors for the synthesis of other important compounds, such as quinazolines and fluorenones. These compounds feature an amino functionality flanked between two nitrile substituents, which makes them valuable for various applications in medicinal chemistry and materials science. The study focuses on developing an efficient and convenient procedure for their synthesis through a carbanion-induced ring transformation of 2H-pyran-2-ones using malononitrile as a carbanion source. This method allows for the creation of a benzene ring at room temperature under mild conditions, without the need for organometallic reagents or catalysts. The synthesized 2-amino-isophthalonitriles are characterized by spectroscopic analysis, confirming their structures and the successful implementation of the synthetic strategy.