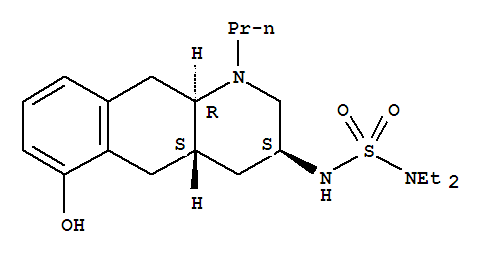
- Chemical Name:Quinagolide
- CAS No.:87056-78-8
- Molecular Formula:C20H33 N3 O3 S
- Molecular Weight:395.62
- Hs Code.:
- Nikkaji Number:J402.834A
- Wikipedia:Quinagolide
- Wikidata:Q4860568
- NCI Thesaurus Code:C73310
- Metabolomics Workbench ID:152151
- ChEMBL ID:CHEMBL290962
- Mol file:87056-78-8.mol
Synonyms:CV 205-502;CV-205-502;CV205-502;N,N-diethyl-N'-(1,2,3,4,4a,5,10,10a-octahydro-6-hydroxy-1-propyl-3-benzo(g)quinolinyl)sulfamide hydrochloride, (3alpha,4aalpha,10abeta)-(+)-isomer;N,N-diethyl-N'-(1,2,3,4,4a,5,10,10a-octahydro-6-hydroxy-1-propyl-3-benzo(g)quinolinyl)sulfamide, (3alpha,4aalpha,10abeta)-(+-)-isomer;Norprolac;quinagolide;quinagolide hydrochloride;quinagolide hydrochloride, (3alpha,4aalpha,10abeta)-(+)-isomer;quinagolide hydrochloride, 3alpha,4aalpha,10abeta-(-)-isomer;quinagolide, (3alpha,4aalpha,10abeta)-(+-)-isomer