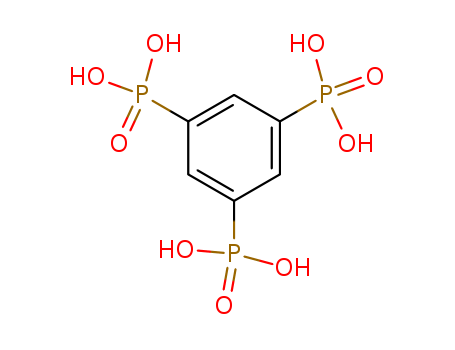
- Chemical Name:(3,5-Diphosphonophenyl)phosphonic acid
- CAS No.:4672-29-1
- Molecular Formula:C6H9O9P3
- Molecular Weight:318.053
- Hs Code.:
- European Community (EC) Number:807-504-7
- Nikkaji Number:J1.751.846A
- Mol file:4672-29-1.mol
Synonyms:(3,5-diphosphonophenyl)phosphonic acid;4672-29-1;benzene-1,3,5-triyltris(phosphonic acid);SCHEMBL20379784;Benzene-1,3,5-trisphosphonic acid;YSZC888;1,3,5-benzenetriphosphonic acid;benzene-1,3,5-triphosphonic acid;MFCD22200395;(3,5-diphosphonophenyl)phosphonicacid;AKOS017345052;CS-0183001;F76987