10.1007/BF02142154
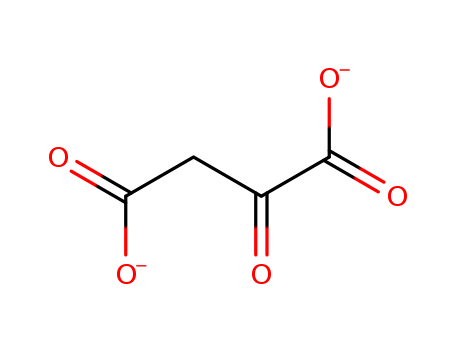
The study investigates the effects of K and Mg aspartate on cellular metabolism, specifically focusing on oxygen consumption and respiratory CO? production in isolated rat nephrons. The chemicals involved include aspartate, which is proposed to stimulate cellular metabolic processes by potentially acting as an anaerobic generator of CO? or stimulating the tricarboxylic acid cycle after deamination. The study found that the addition of aspartate significantly increased oxygen consumption and CO? production without altering the respiratory quotient, suggesting that aspartate enhances cellular metabolism through its role in producing oxalacetate, a key component of the Krebs cycle. This effect is more complex than simply acting as a cation carrier for K and Mg, and may also involve a vascular action due to the vasodilating effect of oxalacetate on small vessels.