10.1016/S0040-4020(97)10286-1
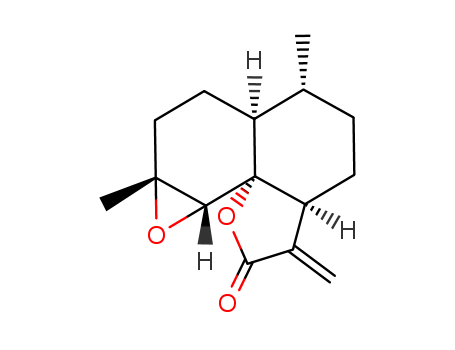
The research details the first-time synthesis of (+)-Artemisinin and (+)-Deoxoartemisinin from Arteannuin B and Arteannuic Acid. The purpose of this study was to develop a short and efficient synthetic route leveraging prior art for the final photo-oxygenation/cyclization reaction, addressing the urgency in discovering novel antimalarial agents due to the emergence of chloroquine-resistant strains of Plasmodium falciparum. The researchers successfully established a synthetic link between Arteannuin B and Artemisinin, as well as a new route from readily available Arteannuic Acid, utilizing a novel oxidative lactonization reaction and a regioselective protection method. The study concluded that the yields for the photo-oxygenation reaction leading to Artemisinin were comparable to previous syntheses, with no observed "ene" products, likely due to the absence of axial allylic protons in their substrates. The yield for the photo-oxygenation to form Deoxoartemisinin was exceptionally high at 65%, attributed to the enhanced rate of cyclization and the absence of competing ene reactions. Key chemicals used in the process included Arteannuin B, Arteannuic Acid, singlet oxygen, Rose Bengal as a sensitizer, and various reagents for protection and deprotection steps, such as 1,2-bis(trimethylsilyloxy)ethane (BTSE), trimethylsilyltriflate (TMSOTf), and lithium aluminum hydride.
10.1016/S0040-4020(03)00330-2
The research focuses on the stereoselective reduction of arteannuin B and its chemical transformations, with the aim of establishing the absolute stereochemistry of dihydroarteannuin B, a derivative of arteannuin B. The study utilized various chemicals and reagents, including Ni2B, NaBH4, CdCl2–Mg–MeOH–H2O, BF3·Et2O–Ac2O, HBr, Ac2O, pyridine, Pd–C, KOH–MeOH, and diglyme, to perform a series of chemical reactions and transformations. The absolute stereochemistry of dihydroarteannuin B was confirmed through 2D NMR and single crystal X-ray diffraction studies. The research concluded that reduction of arteannuin B with Ni2B, NaBH4, or CdCl2–Mg–MeOH–H2O yields dihydroarteannuin B, in which the relative stereochemistry of the C-13 methyl group has been established. The study also discussed experiments aimed at synthesizing dihydrodeoxyarteannuin B, an isomer of dihydroarteannuin B, and its potential transformation to artemisinin, a biologically active component used in antimalarial treatments.