- Chemical Name:Phenylselenenyl chloride
- CAS No.:5707-04-0
- Molecular Formula:C6H5ClSe
- Molecular Weight:191.519
- Hs Code.:29319090
- European Community (EC) Number:227-196-2
- DSSTox Substance ID:DTXSID00205679
- Nikkaji Number:J81.084C
- Wikidata:Q83079302
- ChEMBL ID:CHEMBL3335712
- Mol file:5707-04-0.mol
Synonyms:5707-04-0;Phenylselenenyl chloride;Phenylselenyl chloride;Benzeneselenenyl chloride;phenyl selenohypochlorite;Benzeneselenyl chloride;Chloroselenobenzene;PhSeCl;(phenylselanyl)chlorane;CHEMBL3335712;EINECS 227-196-2;Phenylselenium chloride;Chlorophenylselenium;Phenylselenium monochloride;phenylselenenylchloride;chloroselanylbenzene;Phenylseleno chloride;MFCD00000478;phenylselanyl chloride;C6H5ClSe;phenyl selenium chloride;phenyl selenenyl chloride;C6-H5-Cl-Se;SCHEMBL29416;Phenylselenyl chloride, 98%;WJCXADMLESSGRI-UHFFFAOYSA-;DTXSID00205679;AMY18430;selenohypochlorous acid phenyl ester;BDBM50028968;AKOS015840367;CS-0017812;P1091;D78353;EN300-131834;A831301;Z1255485131


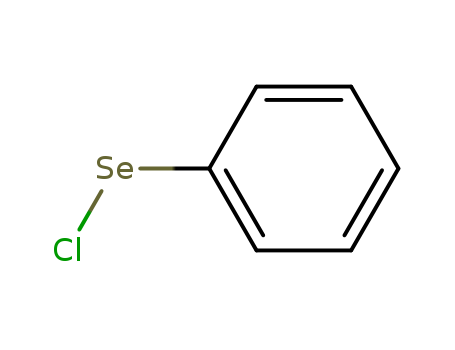
 T;
T;  N
N




