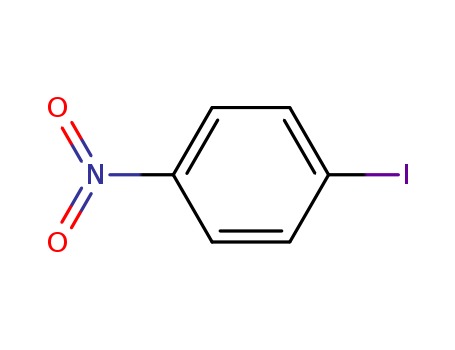
- Chemical Name:1-Iodo-4-nitrobenzene
- CAS No.:636-98-6
- Molecular Formula:C6H4INO2
- Molecular Weight:249.008
- Hs Code.:29049090
- European Community (EC) Number:211-272-7
- NSC Number:9794
- UNII:NH3N3MPW9D
- DSSTox Substance ID:DTXSID7060914
- Nikkaji Number:J47.124K
- Wikidata:Q63399511
- Mol file:636-98-6.mol
Synonyms:1-Iodo-4-nitrobenzene;636-98-6;4-IODONITROBENZENE;Benzene, 1-iodo-4-nitro-;p-Iodonitrobenzene;p-Nitroiodobenzene;4-Nitroiodobenzene;4-iodo-nitrobenzene;p-Nitrophenyl iodide;4-iodo-1-nitrobenzene;4-Nitro-1-iodobenzene;1-iodo-4-nitro-benzene;4-nitrophenyl iodide;30306-69-5;MFCD00007299;4-Iodonitrobenzene-13C6;NSC 9794;NSC-9794;EINECS 211-272-7;AI3-08878;para-nitro iodo-benzene;NSC9794;para-iodonitrobenzene;p-iodo-nitro-benzene;1-Nitro-4-iodobenzene;NH3N3MPW9D;SCHEMBL60337;ghl.PD_Mitscher_leg0.938;1-Iodo-4-nitrobenzene, 98%;DTXSID7060914;SCHEMBL13578687;3-(4-Fluorophenoxy)benzylbromide;BCP14439;STR06257;STK335825;AKOS000120996;AC-22935;AM20040924;FT-0618796;I0064;1-iodo-4-nitro-benzen;4-Iodo-1-nitrobenzene;EN300-17392;A20880;J-504806;Q63399511;Z56924538


 Xn,
Xn, Xi
Xi




