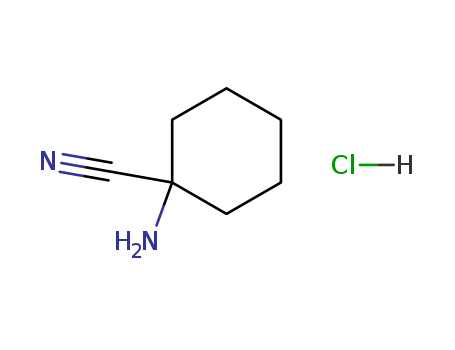
- Chemical Name:1-Aminocyclohexanecarbonitrile hydrochloride
- CAS No.:50846-38-3
- Molecular Formula:C7H12 N2 . Cl H
- Molecular Weight:160.647
- Hs Code.:2926909090
- European Community (EC) Number:256-797-2
- UNII:ZUJ5VG3847
- DSSTox Substance ID:DTXSID50198885
- Mol file:50846-38-3.mol
Synonyms:1-Aminocyclohexanecarbonitrile hydrochloride;50846-38-3;1-cyanocyclohexylamine hydrochloride;Cyclohexanecarbonitrile, 1-amino-, hydrochloride;Cyano-1 amino-1 cyclohexane hydrochloride;1-aminocyclohexanecarbonitrile HCl;1-aminocyclohexane-1-carbonitrile hydrochloride;EINECS 256-797-2;UNII-ZUJ5VG3847;ZUJ5VG3847;1-aminocyclohexane-1-carbonitrile;hydrochloride;Cyclohexanecarbonitrile, 1-amino-, monohydrochloride;1-aminocyclohexanecarbonitrilehydrochloride;SCHEMBL696355;C7-H12-N2.Cl-H;DTXSID50198885;MFCD01734623;AKOS015847763;CS-10092;LS-56528;TS-02136;CS-0168258;FT-0652963;A828319;CYCLOHEXANECARBONITRILE, 1-AMINO-, HYDROCHLORIDE (1:1)