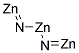Chemical Property of Zinc nitride
Chemical Property:
- Boiling Point:°Cat760mmHg
- Flash Point:°C
- PSA:24.72000
- Density:g/cm3
- LogP:0.35970
- Water Solubility.:Insoluble in water. Soluble in hydrochloric acid.
- Hydrogen Bond Donor Count:0
- Hydrogen Bond Acceptor Count:2
- Rotatable Bond Count:0
- Exact Mass:221.79047
- Heavy Atom Count:5
- Complexity:6.9
- Purity/Quality:
-
98%,99%, *data from raw suppliers
Zinc nitride 99% (metals basis) *data from reagent suppliers
Safty Information:
- Pictogram(s):
- Hazard Codes:
- MSDS Files:
-
SDS file from LookChem
Useful:
- Canonical SMILES:[N-]=[Zn].[N-]=[Zn].[Zn+2]
-
Uses
Zinc nitride (Zn3N2) is a scarcely known II-V compound that crystallizes in the anti-bixbyite structure with a body centered cubic lattice.It presents n-type conductivity due to the substitutional incorporation of oxygen atoms in nitrogen sites. Carrier concentrations in the 1018-1019 cm-3 range have been measured by Hall effect in undoped samples grown by magnetron sputtering and molecular beam epitaxy.2,3 Thin films deposited by radio frequency sputtering present good electrical properties such as high mobility and conductivities in the 10-2-10-3 Ω cm range.
Zinc nitride is a promising candidate for optoelectronics applications due to its high electron mobility and high electrical conductivity. It is also thought that Zn3N2 can be used as a starting material to achieve p-type conductivity in ZnO-based oxide homojunctions. Zinc nitride is useful in optical and electronic devices. It is doped with oxygen to prepare a potential nitride-based transparent conductor, which is used in semiconductors.