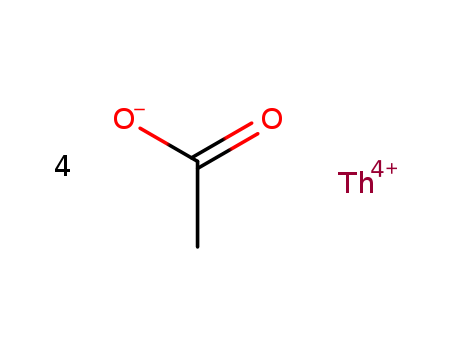
- Chemical Name:Thorium(4+) acetate
- CAS No.:13075-28-0
- Molecular Formula:C2H4 O2 . 1/4 Th
- Molecular Weight:468.217
- Hs Code.:
- European Community (EC) Number:235-972-7
- DSSTox Substance ID:DTXSID90926886
- Mol file:13075-28-0.mol
Synonyms:Thorium(4+) acetate;Acetic acid, thorium(4+) salt;EINECS 235-972-7;13075-28-0;NSC 112213;Acetic acid, thorium(4) salt;C2H4O2.1/4Th;C2-H4-O2.1/4Th;DTXSID90926886;Acetic acid--protactinium (1/1)