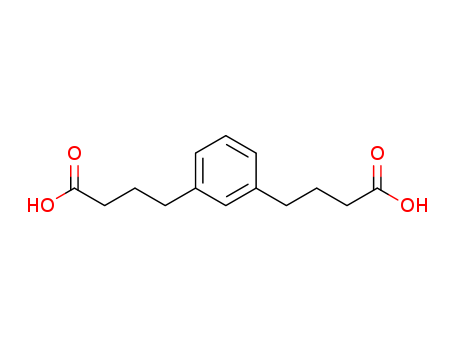
- Chemical Name:1,3-Benzenedibutanoic acid
- CAS No.:54698-75-8
- Molecular Formula:C14H18O4
- Molecular Weight:250.29000
- Hs Code.:
- DSSTox Substance ID:DTXSID80396357
- Nikkaji Number:J1.542.550D
- Wikidata:Q82197024
Synonyms:1,3-Benzenedibutanoic acid;54698-75-8;4-[3-(3-carboxypropyl)phenyl]butanoic Acid;4,4'-(1,3-Phenylene)dibutanoic acid;starbld0044743;Benzene-1,3-dibutanoic acid;DTXSID80396357;A1-32986