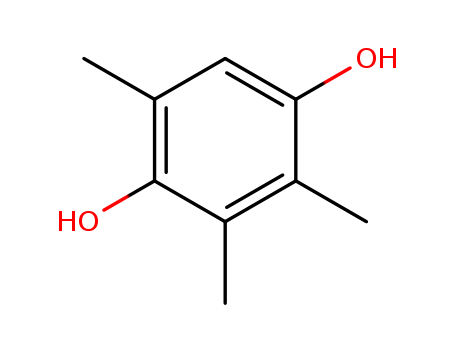
- Chemical Name:Trimethylhydroquinone
- CAS No.:700-13-0
- Molecular Formula:C9H12O2
- Molecular Weight:152.193
- Hs Code.: Oral rat LD50: 3200 mg/kg
- European Community (EC) Number:211-838-3
- NSC Number:401617
- UNII:GSY6340N4A
- DSSTox Substance ID:DTXSID7052446
- Nikkaji Number:J12.289K
- Wikidata:Q27279264
- Metabolomics Workbench ID:130602
- ChEMBL ID:CHEMBL3182864
- Mol file:700-13-0.mol
Synonyms:Hydroquinone,trimethyl- (6CI,7CI,8CI);1,4-Dihydroxy-2,3,5-trimethylbenzene;2,3,5-Trimethyl-1,4-benzenediol;2,3,5-Trimethyl-1,4-hydroquinone;2,3,5-Trimethyl-p-hydroquinone;2,3,5-Trimethylhydroquinone;NSC 401617;Pseudocumohydroquinone;Trimethyl-p-hydroquinone;Trimethylhydroquinone;y-Cumohydroquinone;


 Xn;
Xn;  N
N




