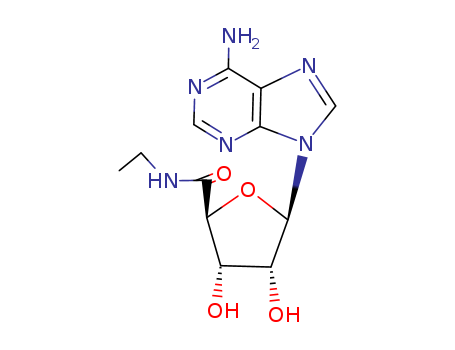
- Chemical Name:5'-N-Ethylcarboxamidoadenosine
- CAS No.:35920-39-9
- Deprecated CAS:100111-00-0,110282-65-0,74992-42-0,84272-21-9
- Molecular Formula:C12H16 N6 O4
- Molecular Weight:317.35
- Hs Code.:29349990
- European Community (EC) Number:636-060-5,636-863-0
- DSSTox Substance ID:DTXSID801017234
- Nikkaji Number:J260.378K
- Wikidata:Q27087892
- Pharos Ligand ID:VWSXGHV42PN5
- Metabolomics Workbench ID:52712
- ChEMBL ID:CHEMBL464859
- Mol file:35920-39-9.mol
Synonyms:5' N Ethylcarboxamidoadenosine;5'-N-Ethylcarboxamidoadenosine;Adenosine 5' N ethyluronamide;Adenosine, N6-Ethyl-carboxamido;Adenosine-5'-(N-ethylcarboxamide);Adenosine-5'-N-ethyluronamide;N Ethylcarboxamidoadenosine;N-Ethylcarboxamidoadenosine;N6 Ethyl carboxamido Adenosine;N6-Ethyl-carboxamido Adenosine;NECA


 T+
T+




