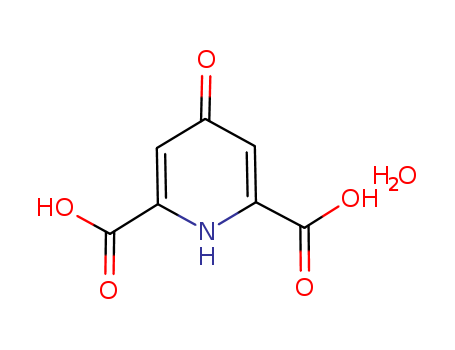
- Chemical Name:4-Oxo-1,4-dihydropyridine-2,6-dicarboxylic acid hydrate
- CAS No.:199926-39-1
- Molecular Formula:C7H5NO5*H2O
- Molecular Weight:201.136
- Hs Code.:2933399090
- DSSTox Substance ID:DTXSID60486644
- Mol file:199926-39-1.mol
Synonyms:199926-39-1;4-Oxo-1,4-dihydropyridine-2,6-dicarboxylic acid hydrate;chelidamic acid monohydrate;4-Oxo-1H-pyridine-2,6-dicarboxylic acid;hydrate;Chelidamic acid hydrate;4-HYDROXYPYRIDINE-2,6-DICARBOXYLIC ACID HYDRATE;2,6-Pyridinedicarboxylic acid, 1,4-dihydro-4-oxo-, hydrate (1:1);1961195-81-2;SCHEMBL113302;Chelidamic acid monohydrate,98;DTXSID60486644;MFCD00150943;MFCD03818117;AKOS015998637;CHELIDAMIC ACID MONOHYDRATE, 98;AS-50193;Chelidamic acid hydrate, >=95%, powder;SY233437;CS-0062895;EN300-207708;O11277;A879902;4-hydroxypyridine-2,6-dicarboxylic acid monohydrate;4-Oxo-1,4-dihydropyridine-2,6-dicarboxylic acid monohydrate;Chelidamic acid hydrate, >=97.0% (dried material, T), ~1 mol/mol water