10.1016/S0040-4039(01)02301-2
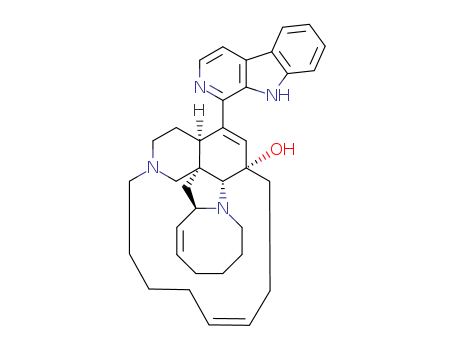
The study focuses on the stereoselective synthesis of the tricyclic core ABC-rings of nakadomarin and manzamine, which are complex marine alkaloids with cytotoxic and antimicrobial properties. The researchers used a common intermediate and the Pauson–Khand cyclization reaction to construct the core structure. Key chemicals involved in the study include enamide 9, cyclopentenone 10, and nakadomarin 1. The Pauson–Khand reaction involved the use of Co2(CO)8 (cobalt carbonyl), n-BuSMe (n-butylthiomethyl ether), and various solvents such as trifluoroethanol and 1,2-dichloroethane. These chemicals served to facilitate the cyclization and subsequent reduction steps, ultimately leading to the formation of the desired tricyclic core structures, which are precursors to both nakadomarin and manzamine. The study also explored the stereochemistry of the products and attempted ring expansion reactions to further synthesize manzamine.