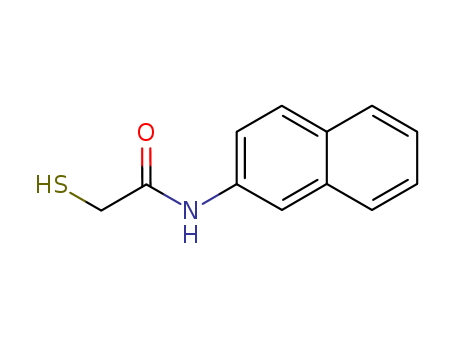
Chemical Property of Thionalide
Chemical Property:
- Vapor Pressure:1.41E-08mmHg at 25°C
- Melting Point:111-112°
- Refractive Index:1.6000 (estimate)
- Boiling Point:458.1°C at 760 mmHg
- Flash Point:230.9°C
- PSA:67.90000
- Density:1.273g/cm3
- LogP:2.78110
- Solubility.:Sparingly soluble in water, readily soluble in organic solvents. The solubility
in water is considerably increased by addition of relatively small amounts
of ethanol or acetic acid.
- XLogP3:2.7
- Hydrogen Bond Donor Count:2
- Hydrogen Bond Acceptor Count:2
- Rotatable Bond Count:2
- Exact Mass:217.05613515
- Heavy Atom Count:15
- Complexity:232
- Purity/Quality:
-
98%min *data from raw suppliers
2-MERCAPTO-N-(2-NAPHTHYL)ACETAMIDE Aldrich *data from reagent suppliers
Safty Information:
- Pictogram(s):
- Hazard Codes:
- MSDS Files:
-
SDS file from LookChem
Useful:
- Chemical Classes:Other Classes -> Naphthalenes
- Canonical SMILES:C1=CC=C2C=C(C=CC2=C1)NC(=O)CS
-
Uses
As a reagent for copper, mercury, silver, thallium, and bismuth. Thionalide reacts with a variety of metal ions to give chelate complexes which
are sparingly soluble in water. The difference in stability of complexes formed by
the reagent with various metal ions in some cases assures a certain analytical
selectivity.
1. Copper(II), silver(I), mercury(II), bismuth(III), arsenic(III), tin(IV), gold(I),
platinum(IV) and palladium(II) give precipitates with the reagent in acidic media
(mineral acids).
2. The copper(II), mercury(II), cadmium(II), thallium(I) and gold(I) complexes
precipitate from alkaline solutions containing tartrate.
3. Thallium(I), antimony(III) and bismuth(III) complexes are precipitated by
the reagent from solutions containing cyanide and tartrate.
4. The thallium(I) complex also precipitates from alkaline (sodium hydroxide)
solution containing cyanide and tartrate.
The above classification shows that the reaction can be made specific for
thallium by appropriate choice of the pH of the solution and application of
suitable masking agents. Similarly, the selectivity of the precipitation reaction can be increased for other ions too, by application of other masking agents. For
instance, tin(IV) can be masked selectively in the presence of arsenic(III) and
antimony(III) with phosphoric acid, while these latter two ions can be precipitated
with thionalide.
The analytical application of thionalide is stimulated by the fact that its metal
complexes are of stoichiometric composition, and can be dried at 105-110°C
and weighed directly.
The reagent may also be utilized in volumetric analytical procedures. The thionalide
content, which is equivalent to the metal to be determined, can be titrated
directly iodometrically, according to the equation:
2C10H7-NH-CO-CH2-SH+I2=C10H7-NH-CO-CH2-S-S-CH2-CO-NH-C10H7+2HI.