10.1016/j.mencom.2020.05.014
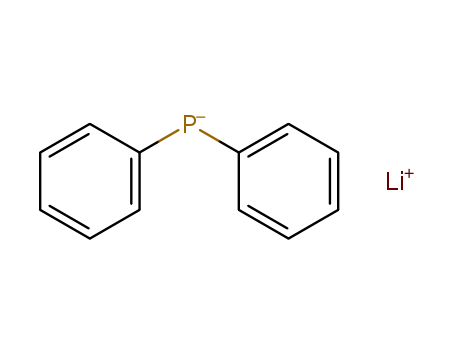
The research focuses on the development of an efficient one-pot synthesis method for diphenyl(pyrazin-2-yl)phosphine, a ligand of interest for its potential use in molecular complexes and coordination polymers due to its catalytic performance, luminescent properties, biological activity, and magnetic features. The synthesis involves the reaction of 2-chloropyrazine with lithium diphenylphosphide (Ph2PLi), which is generated in situ from triphenylphosphine (Ph3P) and metallic lithium in THF. The process also includes the quenching of by-produced PhLi with Me3SiCl. The resulting diphenyl(pyrazin-2-yl)phosphine was then used to form complexes with Ag, Au, and Pt centers, revealing a P-monodentate ligation pattern in the synthesized complexes: [Ag4Cl4(1)4], [Au(1)Cl], and cis-[Pt(1)2Cl2]. The study contributes to the coordination chemistry of P,N-ligands and the synthesis of organophosphorus compounds, providing a new potential pre-catalysts and luminescent materials.