10.1016/s0022-328x(97)00456-7
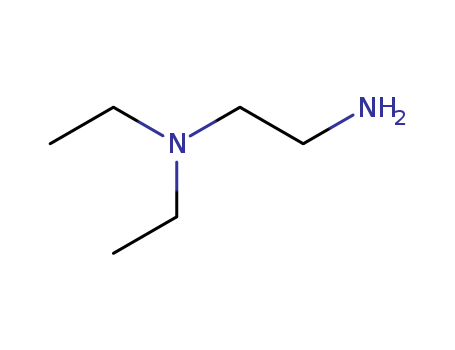
The study investigates the formation of dimeric and trimeric molybdenum(II) complexes containing 2-substituted 3-bonded butadienyl bridging ligands. The starting material used is [MoCI(CO)2(@-CH2(COCI)C=CH2)phen] (phen = 1,10-phenanthroline) (1). When 1 reacts with 1,2-ethanediol or N,N'-diethylethylenediamine in a 2:1 mole ratio, dimeric complexes [MoCI(CO)2(@-CH/(COACH2)C=CH2)phen]2 are formed, where A represents the substituent group (A = O for ester, A = NEt for amide). Reactions with hydroquinone or 1,4-phenylenediamine yield monomeric complexes [MoCI(CO)2(@-CH2(COA)C=CH2)phen], while dimeric complexes are isolated from reactions involving 4,4'-ethylenedianiline or p-xylylenediamine. Attempts to prepare a novel complex bridged by three linked amide substituted butadienyl groups using diethylenetriamine were unsuccessful. However, reaction of 1 with triethanolamine or tris(2-aminoethyl)amine in a 3:1 mole ratio gives trimeric complexes [MoCI(CO)2(~/a-CH2(COACH2CH2)C=CH2)phen]3 N (A = O, NH) in good yield. The study establishes conditions for the formation of these complexes and examines the boundaries of dimer and trimer formation using various bifunctional and trifunctional reagents.


 C,
C,  Xi
Xi



