10.1021/ja027311p
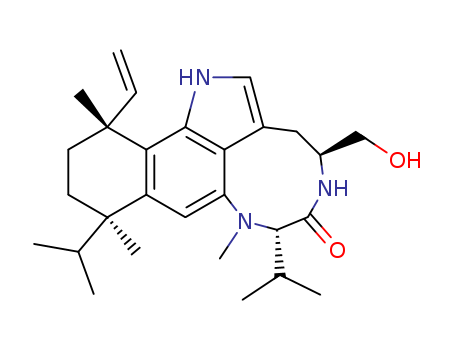
The research aims to develop a novel synthetic method for constructing complex organic molecules by forming C-C bonds through C-H activation, thereby reducing the need for multiple reactive functional groups in coupling reactions. The study focuses on synthesizing the core structure of teleocidin B4, a natural product with significant biological activity, using a series of C-C bond-forming steps that involve C-H bond functionalization. Key chemicals used include tert-butyl aniline derivatives, Pd(II) salts (specifically PdCl2) for cyclometalation, NaOAc as a base, vinyl boronic acids for transmetalation, and methanesulfonic acid for Friedel-Crafts reactions. The researchers successfully synthesized the teleocidin B4 core through a sequence of reactions involving alkenylation, Friedel-Crafts hydroarylation, diastereoselective carbonylation, and intramolecular alkenylation coupling. The final product was obtained in 57% yield after optimization of reaction conditions. The study concludes that C-H bond activation provides a powerful and efficient approach to synthesizing complex natural products, potentially inspiring new chemical transformations and synthetic strategies in organic chemistry.