Refernces
10.1016/S0040-4020(01)97712-9
A study on the oxidation of organic sulfur compounds such as disulfides, thiolsulfinate, thiolsulfonate, thiol, sodium thiolate, and sodium sulfinate using superoxide anion generated from potassium superoxide and 18-crown-6-ether under mild conditions. The research, conducted by Shigeo Oae and colleagues at the University of Tsukuba, demonstrates that these compounds are readily oxidized to both sulfinic and sulfonic acids. The study also notes that sulfide and sulfoxide did not react with the superoxide anion. The oxidation reactions were found to be more effective in polar solvents like pyridine and acetonitrile compared to less polar solvents like benzene. The relative reactivities of the compounds were observed in the order: thiolsulfinate > thiolsulfonate > disulfide = sodium thiolate > sodium sulfinate. The study provides insights into the fundamental nature of the reactions of superoxide anion with organic sulfur compounds and discusses the potential involvement of nucleophilic attack and electron transfer processes in these oxidations.
10.1016/j.tetlet.2006.04.097
The research focuses on the synthesis of substituted 3-furan-2(5H)-ones, which are structural motifs found in numerous bioactive natural products. The methodology involves a Diels–Alder sequence using anthracene and maleic anhydride to form a lactone, which upon deprotonation and electrophilic quenching, yields α-substituted lactones. Key reactants include anthracene, maleic anhydride, sodium borohydride, and various electrophiles such as methyl iodide, allyl iodide, butenyl bromide, benzyl bromide, tributyltin chloride, diethyl chlorophosphate, and chlorotrimethylsilane. The experiments utilize techniques like flash vacuum pyrolysis (FVP) to convert alkylated lactones into 3-substituted furan-2(5H)-ones. The study also explores the challenges and limitations of using cyclopentadiene in such reactions and proposes an alternative route to overcome these issues. Analytical techniques such as 13C NMR and IR spectroscopy were employed to confirm the structure and successful functionalization of the synthesized compounds.
10.1139/v00-091
The study focuses on the preparation and characterization of organic-inorganic hybrid materials incorporating diphosphino moieties through the sol-gel process. Aromatic diphosphines, which are rigid molecules with four hydrolysable SiX3 groups (X = OiPr, H), were used to create new hybrid materials. These materials were characterized using solid-state 13C, 29Si, and 31P NMR spectroscopies. The study aimed to investigate the accessibility of the phosphorus atoms within the resulting xerogels, particularly examining their reactivity with H2O2, S8, CH3I, and W(CO)5·THF. The results showed that all phosphorus atoms reacted quantitatively with the smaller reagents but only 20% with the bulkier W(CO)5·THF, indicating that the rigidity of the inorganic network affected the accessibility of the phosphorus centers. This insight is crucial for understanding the properties and potential applications of such hybrid materials in areas where the reactivity and accessibility of specific atoms within a matrix are significant.
10.1016/j.carres.2007.09.006
The research aimed to develop a novel microwave-accelerated method for methylating soluble starch, which is a process traditionally achieved through more time-consuming and complex procedures. The study successfully demonstrated that soluble starch could be fully methylated in a significantly reduced time frame of 4.66 minutes with a 72% yield, using iodomethane and 30% potassium hydroxide under microwave irradiation. The methylated starch was then efficiently hydrolyzed using 60% formic acid followed by 0.05 M sulfuric acid, both under microwave conditions. The partially methylated monosaccharides were separated and identified through preparative paper chromatography and confirmed by their melting points and optical rotations. The study concluded that microwave irradiation is an effective method for starch methylation, offering a rapid and efficient alternative to traditional methods, without the need for an inert atmosphere or catalysts.
10.1016/S0040-4020(98)01096-5
The research aimed to develop a simple, direct, and efficient method for converting thioamides into thioesters, which are activated carboxylic acid derivatives with applications in synthetic chemistry. The study addressed the need for a reliable, multi-gram synthesis of thioesters and proposed a new route from thioamides that is mild, inexpensive, and experimentally straightforward. The method involved warming thioamides in an aqueous tetrahydrofuran (THF) solution containing an alkylating agent, leading to high yields and being scalable to multi-gram levels. The reactions were found to be tolerant of aryl ethers and acetals, and when volatile alkyl halides were used, products of approximately 95% purity could be obtained without chromatographic purification. Key chemicals used in the process included various thioamides, alkyl iodides, dialkyl sulfates, and activated alkyl bromides as alkylating agents, with methyl iodide (MeI), ethyl iodide (EtI), allyl bromide (allyl-Br), and benzyl bromide (BnBr) being specific examples. The study concluded that the developed method was effective for preparing thioesters from thioamides, offering a practical alternative to existing methods.
10.1246/bcsj.36.1272
The study investigates the condensation reactions of isopropylpyrido[3,2-d]tropolones. The researchers explored various reactions involving these compounds. For instance, 8-isopropyl-2-methylpyrido[3,2-d]tropolone and 9-isopropyl-2-methylpyrido[3,2-d]tropolone reacted with benzaldehyde to produce 8-isopropyl-2-styrylpyrido[3,2-d]tropolone (VIa) and 9-isopropyl-2-styrylpyrido[3,2-d]tropolone (VIb) respectively. The compounds Va, Vb, and Vc, which are derivatives of isopropylpyrido[3,2-d]tropolones, reacted with o-phenylenediamine in acetic acid to yield quinoxaline derivatives such as the quinoxalo derivative of 8-isopropylpyrido[3,2-d]tropolone (VIIla) and the quinoxalo derivative of 8-isopropyl-2-methylpyrido[3,2-d]tropolone (VIIIb). Additionally, the reaction of 8-isopropyl-2-methylpyrido[3,2-d]tropolone (XIb) with phenylhydrazine in acetic acid resulted in the formation of yellow crystals (XIIb) with a pyrido[3,2-d]indolo[2,3-b]tropone structure. The study also attempted to prepare quaternary salts of 8-isopropylpyrido[3,2-d]tropolones by reaction with methyl iodide, ethyl iodide, and ethyl p-toluenesulfonate, but these attempts were unsuccessful. The researchers concluded that the condensation products have specific structures based on their observations and spectral data analysis.
10.3390/molecules21111443
The study focuses on the characterization of the O-methyltransferase enzyme JerF, which is involved in the late stages of jerangolid biosynthesis. JerF is unique for its ability to catalyze the formation of a non-aromatic, cyclic methylenolether, a reaction not previously characterized in other O-methyltransferases. The researchers successfully overexpressed JerF in E. coli and utilized cell-free extracts to conduct bioconversion experiments. They also chemically synthesized a range of substrate surrogates to evaluate JerF's catalytic activity and substrate tolerance. The results revealed that JerF has a broad substrate tolerance and high regioselectivity, making it a promising candidate for chemoenzymatic synthesis, particularly for the modification of natural products containing a 4-methoxy-5,6-dihydro-2H-pyran-2-one moiety. The study also highlighted the potential of JerF in introducing specific methylation patterns and its use in biorthogonal coupling reactions, such as click chemistry, for site-specific labeling of biomolecules like DNA, RNA, or proteins.
10.1016/j.ejmech.2006.06.011
The research focuses on the synthesis, antimicrobial activity, and quantitative structure-activity relationship (QSAR) analysis of new ethyl 3,4-dihydro-3-oxo-4,6,7-trisubstituted-2H-1,4-benzoxazine-2-acetate derivatives. The study aimed to develop novel antimicrobial agents to combat drug resistance and improve treatment outcomes for bacterial and fungal infections. The synthesis involved reactions with various o-aminophenols, monoethyl fumaryl chloride, potassium carbonate, and alkylating agents like methyliodide or ethyliodide. The structures of the synthesized compounds were confirmed using IR, 1H NMR, and mass spectral data. The antimicrobial activity was evaluated using the twofold serial dilution technique against different Gram-positive, Gram-negative bacteria, and Candida species, comparing the results with standard drugs. QSAR analysis was performed using multiple regression analysis to predict the lead optimization for antifungal activity against Candida krusei, considering physicochemical parameters and structural indicators. The research was supported by the Research Fund of Ankara University and employed a series of chemical and microbiological assays, along with statistical data processing, to achieve its objectives.
10.1016/S0022-328X(01)01177-9
The study primarily investigates the reactions of Collman's reagent, [NEt4][CH3COFe(CO)4], with methyl iodide (CH3I) and diphenylacetylene in aprotic solvents, focusing on the synthesis of mono- and dinuclear alkenyl ketone iron complexes. The chemicals used include Collman's reagent as the starting iron complex, methyl iodide to generate reactive 'Fe(CO)4' species, and diphenylacetylene as the alkyne component. These reactants serve to explore the formation of new iron-alkenyl complexes, which are assumed to be key intermediates in various organic syntheses involving acetylenic compounds and transition-metal complexes. The purpose of these reactions is to understand the intermediate iron species involved and to synthesize new organometallic compounds with potential applications in catalysis and organic synthesis.
10.1038/162111a0
The study explores a new ketone synthesis method involving the reaction between substituted malonic esters and acid chlorides, producing acylmalonic esters in high yield. It highlights the potential for synthesizing various carbonyl compounds, including ketones, diketones, keto-acids, and others. Additionally, the study challenges previous assertions about carbon tetrachloride's ineffectiveness in extinguishing petrol fires, emphasizing its historical success and the importance of volatility and vaporization in fire extinguishment. Methyl iodide is also discussed as a more volatile alternative for extinguishing fires compared to carbon tetrachloride, with the study noting the significance of proper application techniques for both chemicals.


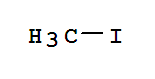
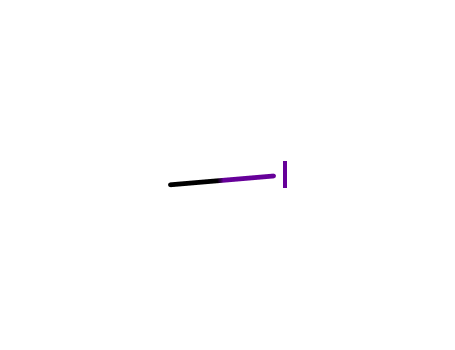
 T,
T,  F,
F,  Xn
Xn
 F:Flammable;
F:Flammable;



