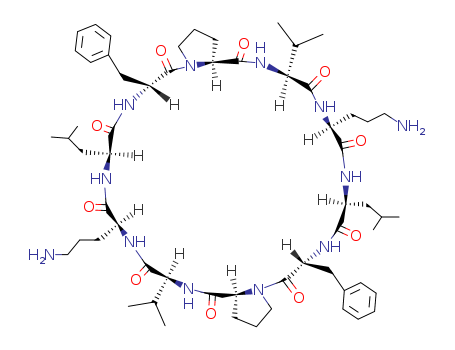
- Chemical Name:GRAMICIDIN C
- CAS No.:113-73-5
- Molecular Formula:C60H92 N12 O10
- Molecular Weight:1141.46
- Hs Code.:
- Mol file:113-73-5.mol
Synonyms:1H,5H-Dipyrrolo[1,2-a:1',2'-p][1,4,7,10,13,16,19,22,25,28]decaazacyclotriacontine,cyclic peptide deriv.; Cyclo(L-leucyl-D-phenylalanyl-L-prolyl-L-valyl-L-ornithyl-L-leucyl-D-phenylalanyl-L-prolyl-L-valyl-L-ornithyl);Cyclo(L-valyl-L-ornithyl-L-leucyl-D-phenylalanyl-L-prolyl-L-valyl-L-ornithyl-L-leucyl-D-phenylalanyl-L-prolyl);Gramicidin C; Gramicidin C (Russian); Gramicidin S 1; Gramicidin S-A; Grammidin