10.1016/S0040-4020(97)00377-3
The study presents an enantioselective synthetic route to trans-2,6-disubstituted piperidines, focusing on the synthesis of (S)-2-methyl tetrahydropyridine-N-oxide, a key intermediate. This compound is crucial for constructing trans-2,6-disubstituted piperidines via a [3+2] nitrone cycloaddition reaction. The research demonstrates the utility of this method by synthesizing the fire ant venom alkaloid, (+)-solenopsin-A, through a series of steps including nitrone formation, cycloaddition, and reductive cleavage. The methodology is highlighted for its potential application in synthesizing similar piperidine-based alkaloids, with implications for pharmaceuticals, such as treatments for Alzheimer's disease.
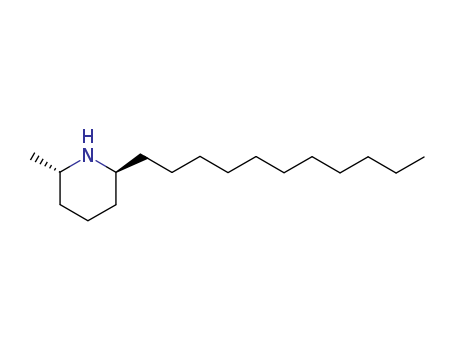
10.1039/c2ob25685a
The research focuses on the total synthesis of piperidine alkaloids, specifically (+)-241D, isosolenopsin, and isosolenopsin A, which are derived from D-alanine. The study employs a gold-catalyzed cyclization as the key step to access the chiral pyridinone intermediates, which are crucial for the synthesis of these alkaloids. The synthesis involves multiple steps, including the preparation of chiral synthons, gold-catalyzed intramolecular cyclization, and stereoselective reductions. The experiments utilized various reactants such as N-Boc-D-alanine, undecyne, BuLi, PPh3AuCl, and AgSbF6, among others, and employed techniques like Arndt–Eistert homologation and catalytic hydrogenation. The synthesized compounds were analyzed using spectroscopic methods (1H and 13C NMR, IR, and HRMS) and optical rotation measurements to confirm their structures and enantiomeric purity.