10.1016/j.tetlet.2004.10.115
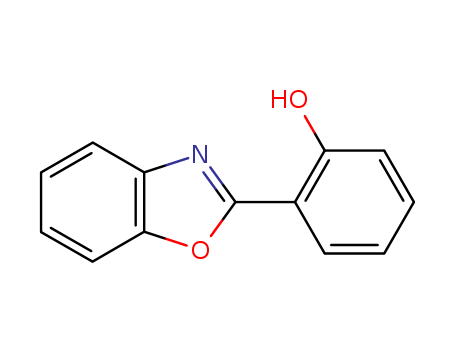
The study presents the design and synthesis of a new fluorescent chelator, Oxa, which possesses two metal-binding sites and is capable of detecting biologically important ions, particularly Zn2+. The chelator was synthesized in six steps and exhibited two distinct dissociation constants for Zn2+, indicating its ability to bind Zn2+ in different concentration ranges. The study also investigated Oxa's affinities for other biologically important ions such as Ca2+ and Mg2+. Key chemicals used in the study include 2-(2'-Hydroxyphenyl)benzoxazole (HBO), known for its excited state intramolecular proton transfer (ESIPT) properties and weak affinity for Zn2+, and aminophenol triacetic acid (APTRA), chosen for its high binding constants and chelating effect. These chemicals served to create Oxa, which combines the properties of both HBO and APTRA to form a molecule with two distinct binding constants for metal ions. The purpose of these chemicals was to develop a fluorescent sensor that could measure ions over a wide concentration range, with a focus on Zn2+ due to its physiological importance.