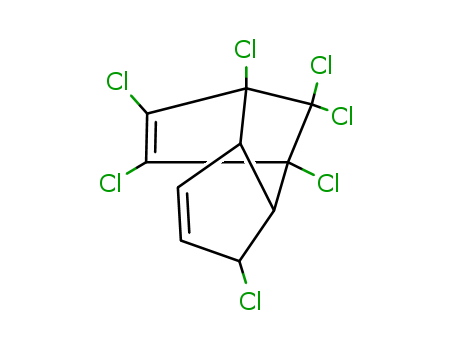
Chemical Property of Heptachlor
Chemical Property:
- Appearance/Colour:white crystalline solid
- Vapor Pressure:0mmHg at 25°C
- Melting Point:95 - 96 C
- Refractive Index:1.5407 (estimate)
- Boiling Point:decomposes
- Flash Point:11oC
- PSA:0.00000
- Density:1.57
- LogP:5.24150
- Storage Temp.:APPROX 4°C
- Water Solubility.:0.056 mg l-1 (25-29 °C)
- XLogP3:4.3
- Hydrogen Bond Donor Count:0
- Hydrogen Bond Acceptor Count:0
- Rotatable Bond Count:0
- Exact Mass:371.818144
- Heavy Atom Count:17
- Complexity:467
- Transport DOT Label:Poison
- Purity/Quality:
-
Heptachlor *data from reagent suppliers
Safty Information:
- Pictogram(s):
Toxic by ingestion, inhalation, and skin absorption; use has been restricted and discontinued except for termite control. TLV: 0.5 mg/m3; animal carcinogen.
- Hazard Codes:T,N,F,Xn
- Statements:
24/25-33-40-50/53-39/23/24/25-23/24/25-11-67-65-38-51/53
- Safety Statements:
36/37-45-60-61-62-16-7
- MSDS Files:
-
SDS file from LookChem
Total 1 MSDS from other Authors
Useful:
- Chemical Classes:Pesticides -> Organochlorine Pesticides
- Canonical SMILES:C1=CC(C2C1C3(C(=C(C2(C3(Cl)Cl)Cl)Cl)Cl)Cl)Cl
- Inhalation Risk:Evaporation at 20 °C is negligible; a harmful concentration of airborne particles can, however, be reached quickly when dispersed, especially if powdered.
- Effects of Short Term Exposure:The substance may cause effects on the central nervous system.
- Effects of Long Term Exposure:The substance may have effects on the liver. This substance is possibly carcinogenic to humans.
-
Description
Heptachlor is a soft, white to light tan, waxy, non-combustible, crystalline solid with a
camphor-like odour. Heptachlor is a member of the cyclodiene group of chlorinated insecticides
(aldrin, dieldrin, endrin, chlordane, heptachlor, and endosulfan) and has a long
history following World War II. It was registered as a commercial pesticide in 1952 for
foliar, soil, and structure applications and for malarial control programmes; after 1960,
it was used primarily in soil applications against agricultural pests and to a lesser extent
against termites.
Heptachlor is available commercially as a dust, a dust concentrate, an emulsifiable concentrate,
a wettable powder, or in oil solutions. It is corrosive to metals and reacts with iron
and rust to form hydrogen chloride gas. Heptachlor is incompatible with many amines,
nitrides, azo/diazo compounds, alkali metals, and epoxides but is stable under normal
temperatures and pressures. It may burn, but does not ignite readily. Heptachlor at high
heat and temperature produces highly toxic, corrosive fumes of hydrogen chlorine gas and toxic oxides of carbon. An important metabolite of heptachlor is heptachlor epoxide
which is an oxidation product formed from heptachlor by many plant and animal species.
Heptachlor is almost insoluble in water but soluble in ether, acetone, benzene, and many
other organic solvents.
-
Physical properties
Colorless to light tan, waxy or crystalline, nonflammable solid with a camphor-like odor
-
Uses
Heptachlor is used for the control of termites, ants, household
insects and soil insects. It is also applied as a seed treatment, soil treatment
or directly to foliage. Insecticide for termite control. Formerly as insecticide for control of cotton boll weevil.