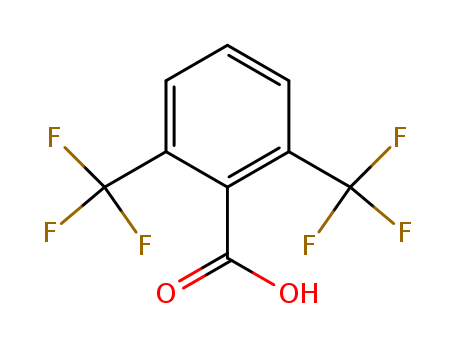
- Chemical Name:2,6-Bis(trifluoromethyl)benzoic acid
- CAS No.:24821-22-5
- Molecular Formula:C9H4 F6 O2
- Molecular Weight:258.12
- Hs Code.:2916399090
- European Community (EC) Number:246-479-1
- UNII:3VW9D8KD6R
- DSSTox Substance ID:DTXSID40179522
- Nikkaji Number:J267.226J
- Wikidata:Q72447186
- Mol file:24821-22-5.mol
Synonyms:2,6-Bis(trifluoromethyl)benzoic acid;24821-22-5;Benzoic acid, 2,6-bis(trifluoromethyl)-;2,6-bis-trifluoromethylbenzoic acid;2,6-di(Trifluoromethyl)benzoic acid;MFCD00000376;3VW9D8KD6R;EINECS 246-479-1;2,6-Bis-trifluoromethyl-benzoic acid;UNII-3VW9D8KD6R;SCHEMBL504216;DTXSID40179522;BBL102248;CK2214;STL556047;AKOS015852700;AC-3899;CS-W010161;PS-11115;2,6-Bis(trifluoromethyl)benzoic acid, 98%;FT-0610519;A19959;EN300-1706142;J-015688;Z276573120