10.1016/j.tetasy.2010.01.007
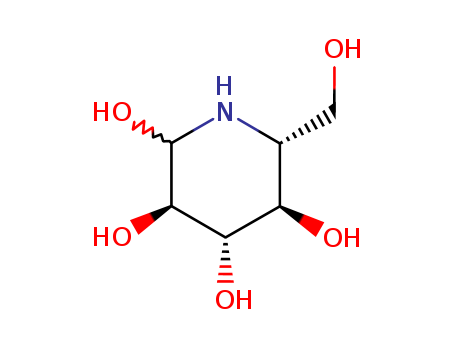
Vrushali H. Jadhav et al. describe a method for synthesizing azepane and nojirimycin iminosugars using the Sharpless asymmetric epoxidation of D-glucose-derived allyl alcohol. The researchers achieved high stereoselectivity in the formation of a- and ?-epoxides, which were then selectively opened with sodium azide to predominantly form 6-azido diols over 5-azido diols. These azido diols were subsequently converted into the corresponding seven-membered azepane and six-membered nojirimycin iminosugars. The study highlights the importance of these iminosugars as glycosidase inhibitors with potential applications in treating diseases like diabetes, cancer, and viral infections. The authors also discuss the challenges and ongoing efforts to improve the regioselectivity of the epoxide ring opening to favor the formation of 5-azido diols, which could further enhance the synthesis of these bioactive compounds.