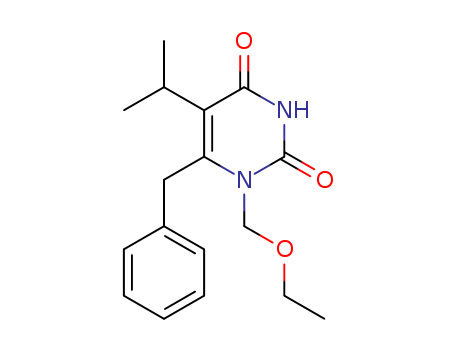
- Chemical Name:Emivirine
- CAS No.:149950-60-7
- Molecular Formula:C17H22 N2 O3
- Molecular Weight:302.373
- Hs Code.:2933990090
- UNII:X87G8IX72O
- DSSTox Substance ID:DTXSID80164437
- Nikkaji Number:J556.890K
- Wikipedia:Emivirine
- Wikidata:Q906062
- NCI Thesaurus Code:C73147
- Metabolomics Workbench ID:56542
- ChEMBL ID:CHEMBL35033
- Mol file:149950-60-7.mol
Synonyms:1-(ethoxymethyl)-5-(1-methylethyl)-6-(phenylmethyl)-2,4(1H,3H)-pyrimidinedione;6-benzyl-1-(ethoxymethyl)-5-isopropyluracil;6-benzyl-1-ethoxymethyl-5-isopropyluracil;emivirine;MKC 442;MKC-442