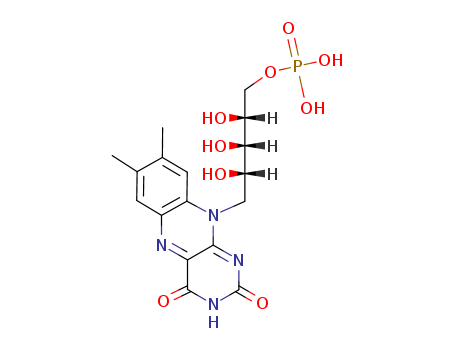
- Chemical Name:Flavin mononucleotide
- CAS No.:146-17-8
- Molecular Formula:C17H21 N4 O9 P
- Molecular Weight:456.349
- Hs Code.:HOSPHATE SODIUM PRODUCT IDENTIFICATION
- European Community (EC) Number:205-664-7
- UNII:7N464URE7E
- DSSTox Substance ID:DTXSID8023559
- Nikkaji Number:J30.175B
- Wikipedia:Flavin_mononucleotide
- Wikidata:Q376061
- NCI Thesaurus Code:C61925
- RXCUI:1925821
- Metabolomics Workbench ID:37845
- ChEMBL ID:CHEMBL1201794
- Mol file:146-17-8.mol
Synonyms:5'-Monophosphate, Riboflavin;5'-Phosphate, Riboflavin;Flavin Mononucleotide;Flavin Mononucleotide Disodium Salt;Flavin Mononucleotide Monosodium Salt;Flavin Mononucleotide Monosodium Salt, Dihydrate;Flavin Mononucleotide Sodium Salt;FMN;Mononucleotide, Flavin;Mononucleotide, Riboflavin;Phosphate, Sodium Riboflavin;Riboflavin 5' Monophosphate;Riboflavin 5' Phosphate;Riboflavin 5'-Monophosphate;Riboflavin 5'-Phosphate;Riboflavin Mononucleotide;Riboflavin Phosphate, Sodium;Sodium Riboflavin Phosphate