Chemical Property of Leflunomide
Chemical Property:
- Appearance/Colour:off white crystalline solid
- Melting Point:163-168 °C
- Boiling Point:289.3 °C at 760 mmHg
- PKA:10.8(at 25℃)
- Flash Point:128.8 °C
- PSA:55.13000
- Density:1.392 g/cm3
- LogP:3.32710
- Storage Temp.:2-8°C
- Solubility.:Practically insoluble in water, freely soluble in methanol, sparingly soluble in methylene chloride.
- XLogP3:2.5
- Hydrogen Bond Donor Count:1
- Hydrogen Bond Acceptor Count:6
- Rotatable Bond Count:2
- Exact Mass:270.06161202
- Heavy Atom Count:19
- Complexity:327
- Purity/Quality:
-
99%Min *data from raw suppliers
Leflunomide *data from reagent suppliers
Safty Information:
- Pictogram(s):
 Xn,
Xn,  Xi
Xi
- Hazard Codes:Xn,Xi
- Statements:
22-36/37/38
- Safety Statements:
26-36
- MSDS Files:
-
SDS file from LookChem
Useful:
- Drug Classes:Antirheumatic Agents
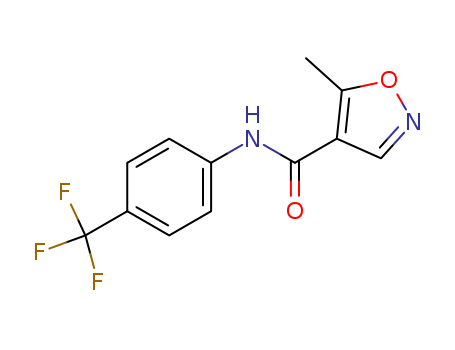
- Canonical SMILES:CC1=C(C=NO1)C(=O)NC2=CC=C(C=C2)C(F)(F)F
- Recent ClinicalTrials:Leflunomide for the Treatment of High-Risk Smoldering Multiple Myeloma in African-American and European-American Patients
- Recent EU Clinical Trials:NEw Clinical End-points in patients with primary Sj?gren’s Syndrome (pSS): an Interventional Trial based on stratifYing patients
-
Uses
(1) It is hydrogenated acid dehydrogenase inhibitor with immunosuppressive and anti-inflammatory effects for the treatment of adults with active rheumatoid arthritis.
(2) Nonsteroidal anti-inflammatory drug. An immunosuppressive. Inhibits T and B cell proliferation. Activity is attributed mainly to its metabolite, a malononitrile derivative, which is beleived to inhibit dihydroorotate dehydrogenase as well as several protein tyrosine kinases. Therapeutical By virtue of its immunosuppressant effects, leflunomide has found use in organ transplantation and treatment of rheumatoid arthritis and other autoimmune diseases.
-
Production method
Ethyl acetoacetate, triethyl orthoformate and acetic anhydride are refluxed together to the complete reaction of raw materials, about 5h. Distillate and collect? 140 150 ℃/1.87kPa fraction, the compound ( I) is obtained, in 85% yield.
Compound (I) is dissolved in ethanol,the mixture of hydroxylamine hydrochloride, sodium acetate and water is added dropwise at 10-15 ℃ and 1h, After dropping,react for about 8h.Concentrated hydrochloric acid and glacial acetic acid are added ,then reflux? for 5h. Concentrate under reduced pressure to 1/2 volume, cool to 10 ℃, filtration, recrystallize ethanol-water? ,compound (Ⅱ) is obtained, it is white crystalline powder, melting point 145-146.5 ℃, in 80% yield.
Compound (II) is dissolved in toluene and thionyl chloride is added dropwise at 50-55℃, reflux for 3h. After concentration, distillation, collect? 78-79 ℃/1.87kPa fraction, the compound (III) is obtained, in 75% yield.
Trifluoromethyl aniline and triethylamine are dissolved? in dichloromethane, at 0-5 ℃ solution ,the compound (Ⅲ) is added. Then at 25--30 ℃ reaction lasts 3h. Water is added, the organic layer is separated, wash with water, wash with brine, and dry. Concentrate under reduced pressure to 1/3 volume, petroleum ether is added, cool, filter, and recrystallize from ethyl acetate to give white crystals powder of leflunomide , m.p. 166~167 ℃, in yield 81%.
-
Description
Leflunomide is an orally-available disease-modifying antirheumatic drug
and was launched as Arava in the US for the treatment of rheumatoid arthritis
(RA) ; it is the first and only drug to be indicated to slow down structural joint
damage of RA, so addressing an unmet medical need.
Leflunomide is prepared in 3 steps from the appropriate acetoacetic anilide
using a nitrile oxide- enamine cycloaddition reaction to assemble the isoxazole
ring. Leflunomide is a prodrug, being extensively metabolized in vivo into the
corresponding 2-cyano-3-hydroxy-2-butenamide resulting from fragmentation of
the isoxazole ring. This cyanoenol is actually the active metabolite and several
experiments in animals have demonstrated that after oral administration,
substantial and sustained levels of this metabolite were delivered to the
systemic circulation.
In vitro, Leflunomide’s active metabolite inhibits dihydroorotate dehydrogenase,
an enzyme involved in the biosynthesis of pyrimidine nucleotides, probably
accounting for its immunosuppressive effect in vivo. Other mechanisms of action
such as inhibition of tyrosine kinase and inhibition of responsiveness to
interleukin-2 have been proposed. In diverse models of autoimmune or allergic
diseases, Leflunornide showed efficacy both prophylactically and therapeutically.
-
Indications
Leflunomide (Arava) is an isoxazole derivative approved
for the treatment of rheumatoid arthritis in
1998. Limited data suggest that it is comparable in efficacy
to sulfasalazine and produces fewer adverse effects.
It has a faster onset of action (4 weeks) than other
DMARDs.
-
Therapeutic Function
Immunosuppressive, Antiarthritic
-
Biological Functions
Leflunomide is inactive, but teriflunomide inhibits pyrimidine de novo synthesis at low therapeutic doses by inhibiting
dihydroorotate dehydrogenase (the rate-determining enzyme for the synthesis of UMP), decreasing DNA and RNA
synthesis, and arresting the cell proliferation cycle and production of antibodies. The reduction of dihydroorotate to
orotate occurs concurrently with the reduction of its cofactor, ubiquinone (coenzyme Q). The inhibition of
dihydroorotate dehydrogenase by teriflunomide demonstrates noncompetitive and uncompetitive kinetics.
Administration of leflunomide in patients with rheumatoid arthritis results in progressive removal of B cells and
down-regulation of the immune process. Teriflunomide not only inhibits B-cell proliferation but also T-cell
proliferation, blocking the synthesis of immunosuppressive cytokines. At high therapeutic doses, leflunomide inhibits
protein tyrosine kinases.
-
Clinical Use
Leflunomide is a DMARD with anti-inflammatory and immunosuppressive activity used for the management of
rheumatoid arthritis. It retards structural damage associated with arthritis in adults who have moderate to severe
active rheumatoid arthritis. Leflunomide also is being investigated for use in patients with solid tumors and organ
transplant recipients.
-
Drug interactions
Potentially hazardous interactions with other drugs
Hepatotoxic or haemotoxic drugs: increased risk of
toxicity.
Cytotoxics: risk of toxicity with methotrexate.
Lipid-lowering agents: effect significantly reduced by
colestyramine - avoid.
Live vaccines: avoid concomitant use.


 Xn,
Xn,  Xi
Xi




