Chemical Property of Carboxymethyl(phosphonomethyl)azanium
Chemical Property:
- Appearance/Colour:white powder
- Melting Point:230 °C
- Refractive Index:1.576
- Boiling Point:465.8 °C at 760 mmHg
- PKA:1.22±0.10(Predicted)
- Flash Point:235.5 °C
- PSA:116.67000
- Density:1.679 g/cm3
- LogP:-0.81330
- Storage Temp.:APPROX 4°C
- Water Solubility.:1.2 g/100 mL
- XLogP3:-4.6
- Hydrogen Bond Donor Count:4
- Hydrogen Bond Acceptor Count:5
- Rotatable Bond Count:4
- Exact Mass:170.02183439
- Heavy Atom Count:10
- Complexity:162
- Purity/Quality:
-
≥98% *data from raw suppliers
Glyphosate *data from reagent suppliers
Safty Information:
- Pictogram(s):
 Xi,
Xi, N,
N, Xn
Xn
- Hazard Codes:Xi,N,Xn
- Statements:
41-51/53-62-37/38-36/37/38-36-22
- Safety Statements:
26-39-61-2-37-36
- MSDS Files:
-
SDS file from LookChem
Total 1 MSDS from other Authors
Useful:
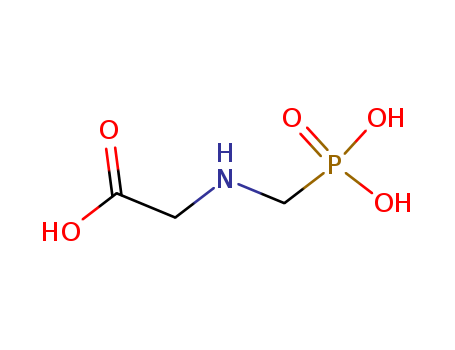
- Canonical SMILES:C(C(=O)O)[NH2+]CP(=O)(O)O
-
Uses
It was originally used for controlling of grass weeds in rubber plantations and can allow the rubber tapping a year earlier and increase the production capacity of the old rubber tree. It is currently gradually extended to forestry, orchards, mulberry fields, tea plantations, rice and wheat, and rape rotation land. Different kinds of weeds have different sensitivity to glyphosate and therefore the dosage is also different. For example, for weeds such as barnyardgrass, green bristlegrass, Alopecurus aequalis, Eleusine indica, crabgrass, cleavers and other annual weeds, the dosage calculated according to the amount of the active ingredient should be 6~10.5 g/ 100 m2. For semen plantaginis, horseweed, and dayflower, the dosage of being an active ingredient should be 11.4~15g/100m2. For cogon, Panicum repens, and reeds, the dosage can be 18~30 g /100m2, generally the used amount of water should be 3~4.5 kg. Apply direct and even spray to the stems and leaves of the weed.
It is a kind of non-selective, post-emergent herbicide with short residue life. It can be used for control of perennial deep roots weeds, annual and biennial weeds, sedges and broadleaf weeds.
Glyphosate is an organic phosphorus herbicide and its herbicidal property was found by D. D. Baird (US) in 1971. Until to the 1980s, it has become an important species in the world herbicide. Herbicide Nonselective, postemergence, broad spectrum herbicide used to control annual and perennial grasses, sedges, broad-leaved and emerged aquatic weeds. This herbicide is also used to control insects on fruit trees. Glyphosate is the active ingredient in several commercial
herbicides. It is a broad-spectrum systemic herbicide for various
types of weeds, grasses (Poaceae), and woody plants.
-
Production method
Dialkyl phosphite ester method
Take glycine, dialkyl phosphite, and paraformaldehyde as raw material and go through addition, condensation, hydrolysis reaction to obtain the product with the purity of 95% and total yield 80% as well as a relative low cost.
Chloromethyl phosphoric acid method
Preparation of chloromethyl phosphoric acid
The phosphorus trichloride and paraformaldehyde were reacted in 200~250 ℃ (corresponding pressure 2.5~3.0 MPa) for 3~5h to obtain the chloromethyl phosphonyl dichloride. It was reported that the ratio is that phosphorus trichloride: polyoxymethylene (1.2~1.5): 1 (mol). In the absence of a catalyst, the yield is 67%. This can be increased to 80% to 89% based on using Lewis acid as the catalyst. The domestic research has not yet reached the level of literature.
Hydrolysis can be obtained chloromethyl phosphoric acid.
Cl2P (O) CH2Cl + H2O → (HO) 2P (O) CH2Cl + HCl
The synthesis of glyphosate: use equal mole of chloromethyl phosphonic acid and glycine, in the aqueous sodium hydroxide (pH> 10), the reaction was refluxed for 10~20h with further acidification to obtain the hydrochloric acid and glyphosate.
If being acidified to a pH 4, that’s monosodium salt; the pH value is 8.5 for disodium salt. If glyphosate was added with equal molar of dimethylamine, giving glyphosate dimethylamine salt.
Iminodiacetic acid method
Preparation of the iminodiacetic acid; the chloroacetic acid, in the presence of calcium hydroxide, is reacted with aqueous ammonia, and undergo acidification, and then sodium hydroxide neutralization to get a yield of 85%.
Or take hydrocyanic acid as raw materials, have it reacted with formaldehyde and ammonia to obtain a yield of 90%.
Preparation of PMIDA The iminodiacetic acid and formaldehyde, phosphorous acid are subject to heating reaction in the presence of sulfuric acid to get PMIDA with the yield being 90%.
Synthesis of glyphosate: PMIDA is mixed with water, with an excess of hydrogen peroxide in the presence of equal molar of sulfuric acid, etc., for heating reaction to obtain glyphosate with a yield of 90% to 95%.
There are many ways for PMIDA oxidation, in addition to hydrogen peroxide; we can also use concentrated sulfuric acid, precious metals (palladium, rhodium, etc.) oxide, and activated carbon air oxidation or electrolysis method. For example, 10 parts of PMIDA, 170 parts of water and 0.6 parts of 5% palladium-charcoal catalyst were placed in an autoclave, put through oxygen to 2.07X105Pa, reacted at 90~100 °C to obtain it with the yield of 96% and purity of 97%.
There are two methods of production from sub-categories, namely, method 1 using iminodiacetic acid (IDA) as raw material and method 2 using glycine-dialkyl phosphite as raw materials, wherein the glycine-dialkyl phosphite method has a largest production capacity, the largest number of production companies, about 80% of the total production of glyphosate production accounts reached 70,000 t; Production capacity of IDA law is nearly 30,000 t. There are four methods in subdivision: 1) homemade (by chloroacetic acid method) IDA, thought the method of PMIDA being subject to oxidation of concentrated sulfuric acid has gained some progress since the 1980s, owing to the large amount of strong calcium chloride acidic wastewater, causing a low yield (IDA yield of about 70%), and can only be made into water preparation with the highest annual output of 2000t (10% water). Upon entering into the 1990s, the amount of the produced company gradually reduced with lower yields. 2) Company of self-production of trimethyl phosphite for the production of glyphosate; this method apply water instead of methanol as solvent, with sodium hydroxide instead of triethylamine as the catalyst which reduces the post-treatment with a yield of 65%. However, owing to the higher price of trimethyl than dimethyl, only companies of self-production of trimethyl can have the cost being with certain market competitiveness. 3) There are many enterprises for applying dimethyl phosphite for glyphosate production; it has large-scale production plant. With optimized production process, advanced equipment and automatic systems, this method has made certain technical progress with the product quality indicators reaching the international market requirements, and therefore China mainly uses this process. 4) Homemade (use diethanolamine for dehydrogenation oxidation) IDA or outsourcing IDA, the use of hydrogen peroxide oxidation of PMIDA for preparation of glyphosate. Fixed material consumption: paraformaldehyde 500 kg/t, methanol 500kg/t, glycine 700 kg/t, triethylamine 50 kg t, dimethyl phosphite 1000 kg/t, hydrochloric acid 3000 kg/t
-
Description
Glyphosate (N-(phosphonomethyl)glycine; 1071-83-6) is the
active ingredient in several commercial herbicides for nonselective
weed control. Glyphosate herbicides are among the
world’s most widely used herbicides. Roundup?, containing
the active ingredient glyphosate, was developed and introduced
by Monsanto Company in 1974. Other formulations include
WeatherMax, UltraMAX, Buccaneer, Razor Pro, Rodeo, and
AquaMaster?. Some crops such as soybeans and cotton have
been genetically engineered to be resistant to glyphosate
(Roundup Ready), allowing farmers to use glyphosate as
a postemergence herbicide. The United States Environmental
Protection Agency (EPA) considers glyphosate to be relatively
low in toxicity compared to organochlorine and organophosphate
pesticides.


 Xi,
Xi, N,
N, Xn
Xn




