- Chemical Name:Vitamin P
- CAS No.:153-18-4
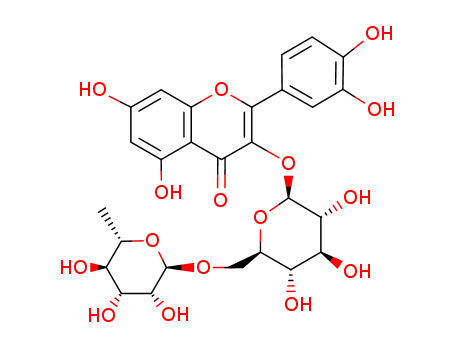
- Molecular Formula:C27H30O16
- Molecular Weight:610.526
- Hs Code.:29381000
- European Community (EC) Number:215-672-2
- NSC Number:408168,9220
- DSSTox Substance ID:DTXSID70859300
- Wikidata:Q27163468
- Metabolomics Workbench ID:130886
- ChEMBL ID:CHEMBL32579
- Mol file:153-18-4.mol
Synonyms:3',4',7-trihydroxyethylrutin;3',4',7-tris(O-(2- hydroxyethyl))rutin;oxerutin;Paroven;Posorutin;Relvene;Rhéoflux;Teboven;Troxérutine Mazal;troxerutin;Troxerutin-ratiopharm;Troxeven;Vastribil;Veinamitol;Veniten retard;Veno SL;Venorutin;venoruton;venoruton P4;Venotrulan Trox;vitamin P4


 Xn,
Xn, Xi
Xi




