- Chemical Name:Aluminium-nickel
- CAS No.:12635-29-9
- Molecular Formula:AlNi
- Molecular Weight:85.67
- Hs Code.:38151900
- European Community (EC) Number:234-439-6,682-897-4,684-239-1
- DSSTox Substance ID:DTXSID0065145,DTXSID10276552
- Mol file:12635-29-9.mol
Synonyms:Aluminium-nickel;12635-29-9;12003-78-0;12635-27-7;aluminum;nickel;nickel aluminum;nickel-aluminum;Aluminum-nickel catalyst;Aluminium nickel, Al/Ni 50/50;Al.Ni;aluminum-nickel;aluminium nickel;nickel aluminium;nickel-aluminium;Nickel aluminide;Niquel Aluminium;Raney nickel aluminium;Nickel Aluminide Powder;Catalyseur d'aluminium-nickel;KSC005Q4L;DTXSID0065145;DTXSID10276552;NPXOKRUENSOPAO-UHFFFAOYSA-N;EINECS 234-439-6;AKOS015833427;AKOS015903659;Aluminium, compound with nickel (1:1);FT-0622245;Nickel aluminum, 99.0% min (metals basis);Raney(R) Nickel Catalyst, 50% slurry in H2O;Aluminum-nickel catalyst, Al-Ni 50:50 wt. %;Raney Nickel (R)2800 slurry, in H2O, active catalyst;Raney(R)-Nickel, W.R. Grace and Co. Raney(R)2400, slurry, in H2O, active catalyst;Raney(R)-Nickel, W.R. Grace and Co. Raney(R)2800, slurry, in H2O, active catalyst;Raney(R)-Nickel, W.R. Grace and Co. Raney(R)3202, slurry, in H2O, active catalyst;Raney(R)-Nickel, W.R. Grace and Co. Raney(R)4200, slurry, in H2O, active catalyst;12704-83-5

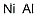

 F,
F,  Xn
Xn


