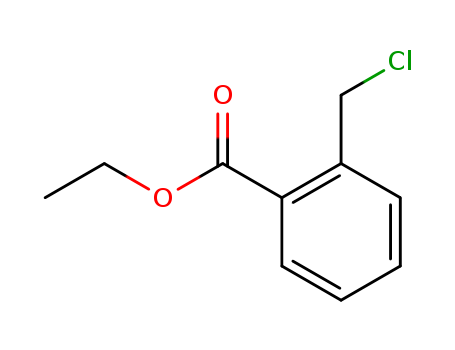
- Chemical Name:Ethyl 2-(chloromethyl)benzoate
- CAS No.:1531-78-8
- Molecular Formula:C10H11ClO2
- Molecular Weight:198.649
- Hs Code.:2916399090
- DSSTox Substance ID:DTXSID30513436
- Nikkaji Number:J33.619J
- Wikidata:Q72473683
- Mol file:1531-78-8.mol
Synonyms:Ethyl 2-chloromethylbenzoate;1531-78-8;ethyl 2-(chloromethyl)benzoate;ETHYLO-(CHLOROMETHYL)BENZOATE;2-(chloromethyl)-benzoic acid ethyl ester;o-(chloromethyl)benzoic Acid Ethyl Ester;ethyl 2-chloromethyl-benzoate;SCHEMBL2825239;DTXSID30513436;QQOVRPBUAUNBAV-UHFFFAOYSA-N;MFCD00053935;AKOS006275395;2-chloromethyl-benzoic acid ethyl ester;AS-12693;FT-0660339;Methanone, 4-morpholinyl(tetrahydro-2-furanyl)-;A921536