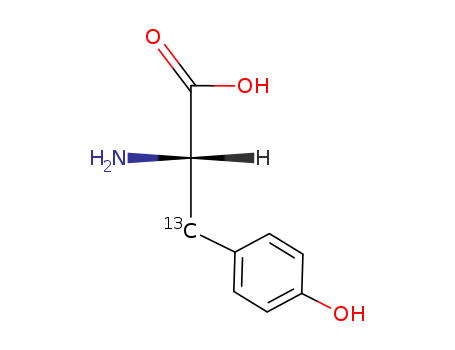
- Chemical Name:L-Tyrosine-3-13C
- CAS No.:129077-96-9
- Molecular Formula:C9H11NO3
- Molecular Weight:182.18
- Hs Code.:
- European Community (EC) Number:693-233-8
- DSSTox Substance ID:DTXSID90480574
- Nikkaji Number:J980.729B
- Wikidata:Q82315444
- Mol file:129077-96-9.mol
Synonyms:L-Tyrosine-3-13C;129077-96-9;(2S)-2-amino-3-(4-hydroxyphenyl)(313C)propanoic acid;L-TYROSINE-BETA-13C;L-Tyrosine-b-13C;DTXSID90480574;L-Tyrosine-3-13C, 99 atom % 13C, 99% (CP)


 Xi
Xi



