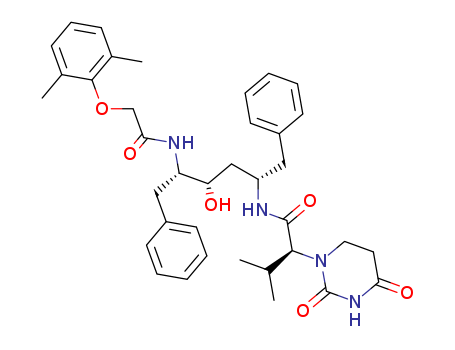
- Chemical Name:Lopinavir Metabolite M-1
- CAS No.:192725-39-6
- Molecular Formula:C37H46 N4 O6
- Molecular Weight:642.792
- Hs Code.:
- Mol file:192725-39-6.mol
Synonyms:1(2H)-Pyrimidineacetamide,N-[(1S,3S,4S)-4-[[(2,6-dimethylphenoxy)acetyl]amino]-3-hydroxy-5-phenyl-1-(phenylmethyl)pentyl]tetrahydro-a-(1-methylethyl)-2,4-dioxo-, (aS)- (9CI);1(2H)-Pyrimidineacetamide, N-[4-[[(2,6-dimethylphenoxy)acetyl]amino]-3-hydroxy-5-phenyl-1-(phenylmethyl)pentyl]tetrahydro-a-(1-methylethyl)-2,4-dioxo-,[1S-[1R*(R*),3R*,4R*]]-