Chemical Property of Rucaparib
Chemical Property:
- Boiling Point:625.248 °C at 760 mmHg
- PKA:14.10±0.20(Predicted)
- Flash Point:331.938 °C
- PSA:56.92000
- Density:1.281 g/cm3
- LogP:3.69900
- Storage Temp.:2-8°C(protect from light)
- Solubility.:DMSO (Slightly), Methanol (Slightly)
- XLogP3:2.5
- Hydrogen Bond Donor Count:3
- Hydrogen Bond Acceptor Count:3
- Rotatable Bond Count:3
- Exact Mass:323.14339037
- Heavy Atom Count:24
- Complexity:466
- Purity/Quality:
-
99%, *data from raw suppliers
Rucaparib *data from reagent suppliers
Safty Information:
- Pictogram(s):
- Hazard Codes:
- MSDS Files:
-
SDS file from LookChem
Useful:
- Drug Classes:Antineoplastic Agents
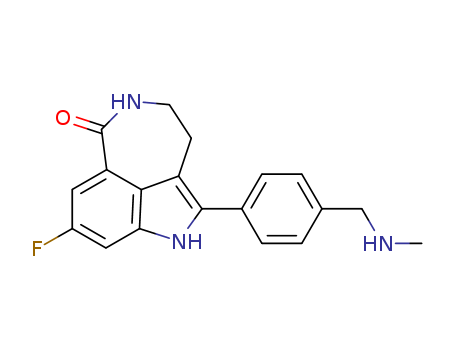
- Canonical SMILES:CNCC1=CC=C(C=C1)C2=C3CCNC(=O)C4=C3C(=CC(=C4)F)N2
- Recent ClinicalTrials:PLX038 (PEGylated SN38) and Rucaparib in Solid Tumors and Small Cell Cancers
- Recent EU Clinical Trials:A randomized phase II trial of Mirvetuximab soravtansine (IMGN853), in folate receptor alpha (FRα) high recurrent ovarian cancer eligible for platinum-based chemotherapy.
- Recent NIPH Clinical Trials:A Study in Ovarian Cancer Patients Evaluating Rucaparib and Nivolumab as Maintenance Treatment Following Response to Front-Line Platinum-Based Chemotherapy (ATHENA)
-
Description
Rucaparib is a PARP inhibitor used as an anti-cancer agent. Rucaparib is a first-in-class pharmaceutical drug targeting the DNA repair enzyme poly-ADP ribose polymerase-1 (PARP-1). It was discovered as part of a collaboration between scientists working at the Northern Institute of Cancer Research and Medical School of Newcastle University and Agouron Pharmaceuticals in San Diego, California. It is being developed by Clovis Oncology. Rucaparib was
approved in the US as an oral treatment for advanced ovarian
cancer. Development of rucaparib began with collaborations
between Cancer Research UK and Agouron Pharmaceuticals
(later acquired by Pfizer). Global development rights for
rucaparib were ultimately granted to Clovis Oncology via a
licensing agreement from Pfizer. To qualify for treatment with
rucaparib monotherapy, patients must demonstrate deleterious
breast cancer (BRCA) mutation (germline and/or somatic)-
associated advanced ovarian cancer and also must have
previously been treated with two or more chemotherapy
regimens. Rucaparib functions as a small molecule poly(ADPribose)
polymerase (PARP) inhibitor, which plays an important
role in DNA repair. This newly approved drug displays
nanomolar potency against PARP-1, -2, and -3 enzymes,
which translates into improved efficacy over alternative
therapies such as olaparib or niraparib. Furthermore,
rucaparib is also known to cause vasodilation, which is thought
to induce tumor perfusion and increased accumulation of the
drug in cancer cells. Although rucaparib shows higher
cytotoxicity in cancer cells with mutation of BRCA1/2 genes
and other DNA repair genes, reduced tumor growth was
observed in mouse xenograft models of human cancers with
and without BRCA mutations.Rucaparib is also being
pursued as a treatment for breast cancer and has displayed
promising initial results in trials for pancreatic cancer. Poly(ADP-ribose) polymerases (PARPs) are activated by DNA single- and double-strand breaks and promote repair of DNA damage through the relaxation of chromatin and recruitment of other repair proteins. Inhibition of PARP activity has been linked to synthetic lethality in cells with mutations in BRCA1 or BRCA2 and is used as a therapeutic strategy to selectively target cancers. Rucaparib is a potent, cell-permeable inhibitor of PARP1 (Ki = <5 nM) that is used in clinical therapy to sensitize cancer cells to chemotherapy. Rucaparib inactivates PARP activity in cells with homologous recombination DNA repair pathway mutations at LC50 values ranging from 1.3-5.5 μM. At 25 mg/kg, rucaparib arrests tumor growth in mice bearing epigenetically silenced BRCA1 UACC3199 xenograft tumors. It has been shown to increase efficacy of temozolomide in medulloblastoma cells and xenografts.
-
Uses
Rucaparib is PARP1 inhibitor. It can be used in biological study of chemical screening to identify drugs that enhance or mitigate cellular responses to antibody-toxin fusion proteins using human B cell precursor leukemia cells and cervical adenocarcinoma cells.