10.1016/0040-4020(82)85150-8
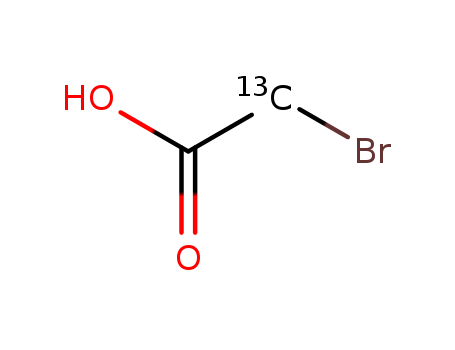
The research aimed to synthesize 13C and 15N labeled (S)-tryptophan, a crucial amino acid used in biosynthetic studies of various compounds and for conformational studies in proteins. The study utilized a biosynthetic approach involving the incubation of (R,S)-serine-1-13C in a culture of Escherichia coli cells, which were engineered to produce (S)-tryptophan-1-13C. Additionally, the researchers synthesized indole-3-13C and indole-15N through known chemical sequences, and these isotopes were then converted by the E. coli to yield (S)-tryptophan-1-13C and (S)-tryptophan-indole-15N, respectively. The chemicals used in this process included (R,S)-serine-1-13C, bromoacetyl bromide-2-13C and 15N-labeled aniline, among others. The conclusions of the study highlighted the efficiency of this biosynthetic method, which could accommodate various labeling patterns and was considered the simplest route for the preparation of optically active products, with yields ranging from 40 to 50%.