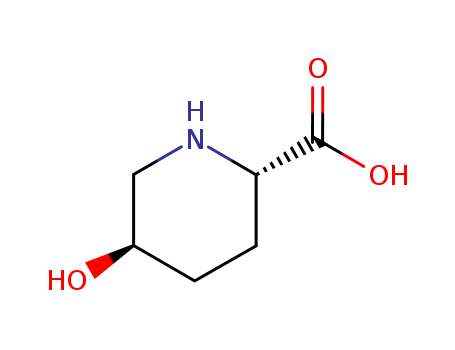
- Chemical Name:(2S,5R)-5-hydroxypiperidine-2-carboxylic acid
- CAS No.:50439-45-7
- Molecular Formula:C6H11NO3
- Molecular Weight:145.158
- Hs Code.:2933399090
- UNII:N38XA45QRV
- DSSTox Substance ID:DTXSID001302217
- Nikkaji Number:J664.550J
- Wikidata:Q105238374
- Mol file:50439-45-7.mol
Synonyms:50439-45-7;(2S,5R)-5-Hydroxypipecolic acid;(2S,5R)-5-hydroxypiperidine-2-carboxylic acid;(2S,5R)-trans-5-Hydroxypiperidine-2-carboxylic acid;trans-5-Hydroxy-L-pipecolic acid;N38XA45QRV;2-Piperidinecarboxylic acid, 5-hydroxy-, (2S,5R)-;L-trans-5-Hydroxypipecolic acid;5-Hydroxy-L-pipecolic acid, trans-;5-Hydroxy-L-pipecolic acid, trans-(-)-;UNII-N38XA45QRV;2-Pyrimidinecarboximidamide HCl;SCHEMBL1250475;DTXSID001302217;AKOS006351657;AMY202003234;CS-0138319;A902404;W-201171