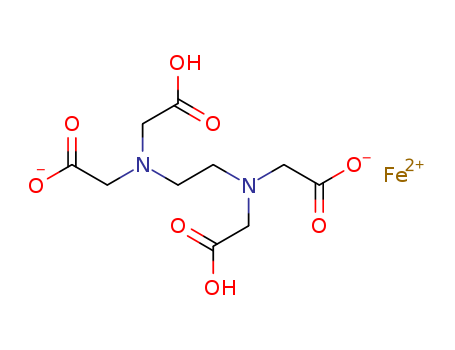
- Chemical Name:Iron(2+) 2,2'-({2-[bis(carboxymethyl)amino]ethyl}azanediyl)diacetate
- CAS No.:21393-59-9
- Molecular Formula:C10H12 Fe N2 O8 . 2 H
- Molecular Weight:346.077
- Hs Code.:
- DSSTox Substance ID:DTXSID60926070
- Nikkaji Number:J136.015I,J136.016G
- Mol file:21393-59-9.mol
Synonyms:DTXSID60926070;JVXHQHGWBAHSSF-UHFFFAOYSA-L;Iron(2+) 2,2'-({2-[bis(carboxymethyl)amino]ethyl}azanediyl)diacetate