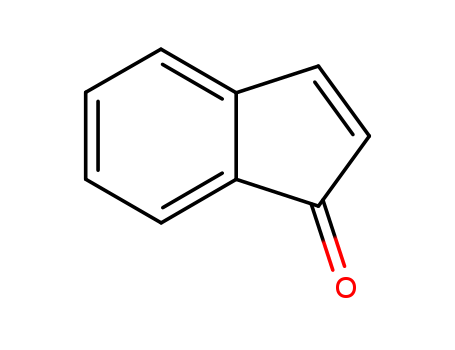
10.1055/s-2005-923587
The study focused on developing a facile method to construct 1H-inden-1-one, a structural motif commonly found in many natural products with pharmacological properties such as cytotoxicity, anti-HIV and antibacterial activities. The aim of the study was to construct multiple chiral centers in one step using the intramolecular Heck reaction. The researchers unexpectedly found that when compound 1 was treated with a catalytic amount of Pd(OAc)2 in the presence of air, only the oxidation product 1H-inden-1-one 2 was obtained. The reaction was found to involve an intramolecular Heck reaction followed by air oxidation of the allylic alcohol. The optimal reaction conditions were determined to be 24 h at 80°C, using 5 mol% Pd(OAc)2 and 15 mol% PPh3, DMF as solvent, and K2CO3 as base. The study concluded that this new method can be used to construct indenone/indanone skeletons, which are important motifs in the synthesis of natural products. The chemicals used in the process include 1-(2-bromoaryl)prop-2-en-1-ol as substrate, Pd(OAc)2 as palladium catalyst, PPh3 as ligand, K2CO3 as base, and DMF as solvent.