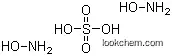10039-54-0 Usage
Chemical Properties
Also hydroxylammonium sulfate, or HS, (NH2OH)2·H2S04 is colorless crystals that are soluble in water and slightly soluble in alcohol. The solution has a corrosive action on the skin. Used as a reducing agent, photographic developer, purification agent for aldehydes and ketones, chemical synthesis, textile chemical, oxidation inhibitor for fatty acids, catalyst, in biological and biochemical research, for making oximes for paints and varnishes, and rustproofing.
Physical properties
Colorless, crystalline solid; melts at 177°C (decomposes); very soluble in386 HYDROXYLAMINE HYDROCHLORIDE / HYDROXYLAMINE SULFATEpp-03-25-new dots.qxd 10/23/02 2:38 PM Page 386 water; slightly soluble in alcohol.
Uses
Different sources of media describe the Uses of 10039-54-0 differently. You can refer to the following data:
1. Hydroxylammonium sulfate is a reducing agent in photography; catalyst, swelling agent, and copolymerization inhibitor in polymerization processes; in chemical
synthesis; as a textile chemieal; as an oxidation inhibitor; in making oximes for paints and varnishes; in rustproofing; in
nondiscoloring short -stoppers for synthetic rubbers; for unhairing hides; in biological and biochemical research; as a purification
agent for aldehydes and ketones; converts aldehydes and ketones to oximes and acid chlorides to hydroxamic acids.
2. Hydroxylamine sulfate may be used to prepare highly sensitive cellulose tape, used for the detection of formaldehyde gas. It may be used in the quantitative determination of perchlorate in biological fluids by spectrophotometric methods .
3. Used as to purify aldehydes and ketones; reagent for mercury and silver detection in water; reducing agent.
4. As reducing agent in photography; in synthetic and analytical chemistry; to purify aldehydes and ketones. As antioxidant for fatty acids and soaps. As dehairing agent for hides.
Preparation
Hydroxylamine sulfate may be prepared by mixing stoichiometric amountsof hydroxylamine and sulfuric acid. It also may be prepared by electrolytical-ly reducing an aqueous solution of ammonium sulfate.
General Description
Colorless crystalline solid. Contact may cause severe irritation to skin, eyes, and mucous membranes. May be toxic by ingestion.
Air & Water Reactions
Water soluble.
Reactivity Profile
Sulfuric acid fumes may form in fires [USCG, 1999]. Solid Hydroxylamine sulfate explodes when heated to 170°C., [Chem. Process 26:30(1963)]. Sodium ignites on contact with hydroxylamine. (Mellor, 1940, Vol. 8, 292.)
Hazard
Irritant to tissue.
Health Hazard
Inhalation of dust or ingestion may cause systemic poisoning characterized by cyanosis, methemoglobinemia, convulsions, and coma. Contact with eyes or skin causes irritation.
Fire Hazard
Special Hazards of Combustion Products: Sulfuric acid fumes may form in fires.
Flammability and Explosibility
Nonflammable
Safety Profile
Poison by skin contact and intraperitoneal routes. Mutation data reported. A corrosive irritant to skin, eyes, and mucous membranes. Moderately explosive when exposed to heat or by chemical reaction. In the presence of alkalies at elevated temperatures, free hydroxylamine is liberated and may decompose explosively. When heated to decomposition it emits toxic fumes of SOx and NOx. See also AMINES and SULFATES.
Purification Methods
Crystallise it from boiling water (1.6mL/g) by cooling to 0o.
Check Digit Verification of cas no
The CAS Registry Mumber 10039-54-0 includes 8 digits separated into 3 groups by hyphens. The first part of the number,starting from the left, has 5 digits, 1,0,0,3 and 9 respectively; the second part has 2 digits, 5 and 4 respectively.
Calculate Digit Verification of CAS Registry Number 10039-54:
(7*1)+(6*0)+(5*0)+(4*3)+(3*9)+(2*5)+(1*4)=60
60 % 10 = 0
So 10039-54-0 is a valid CAS Registry Number.
InChI:InChI=1/2H3NO.H2O4S/c2*1-2;1-5(2,3)4/h2*2H,1H2;(H2,1,2,3,4)