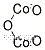1308-04-9 Usage
Uses
Different sources of media describe the Uses of 1308-04-9 differently. You can refer to the following data:
1. COBALT(III) OXIDE BLACK is used as a pigment; for glazing porcelain and pottery; and for coloring enamels.
2. Cobaltic oxide (Co2O3) is also known as cobalt oxide or cobalt black. It is dark gray to black
and is used in pigments, ceramic glazes, and semiconductors.
3. Pigment, coloring enamels, glazing pottery.
Preparation
Different sources of media describe the Preparation of 1308-04-9 differently. You can refer to the following data:
1. COBALT(III) OXIDE BLACK is prepared by heating cobalt compounds at low temperatures in air.
2. Cobalt(III) oxide is prepared by heating cobalt compounds at low temperatures in air.
Reactions
Heating with hydrogen, carbon or carbon monoxide reduces the oxide to cobalt metal.
Co2O3 + 3H2→ 2Co + 3H2O
Co2O3 + 3CO→ 2Co + 3CO
2Co2O3 + 3C→ 4Co + 3CO2
Strong heating in air converts cobalt(III) oxide to tricobalt tetroxide.
Reactions with mineral acids produce their Co3+ salts:
Co2O3 + 6HCl → 2CoCl3 + 3H2O
Description
Neither the oxide Co2O3 nor the hydroxide Co(OH)3 is definitely established. The oxidation of cobalt(II) hydroxide in aqueous suspension with, for example, peroxides, or the destruction of a cobalt(III) complex with alkali gives a brown or black powder Co2O3.aq which upon drying at 150° yields the monohydrate Co2O3.H2O; this is probably best formulated as CoO(OH). When heated further in attempts to dehydrate it, the "monohydrate" begins to evolve oxygen (as well as water) at 300° with the formation of black Co3O4. When heated to 100° in air cobalt(II) hydroxide is converted to dark brown CoO(OH).
Chemical Properties
Steel-gray or black powder.Soluble in concentrated acids; insoluble in water.
Physical properties
Grayish black powder; density 5.18 g/cm3; decomposes at 895°C; insoluble in water; soluble in concentrated mineral acids.
Definition
Sometimes incorrectly called cobalt peroxide.
Safety Profile
: Moderately toxic by
intraperitoneal and subcutaneous routes.
Questionable carcinogen. Violent reaction
with hydrogen peroxide. The oxide increases
the sensitivity of nitroalkanes (e.g.,
nitromethane, nitroethane, and 1
nitropropane) to heat or detonation. See
also COBALT COMPOUNDS.
Check Digit Verification of cas no
The CAS Registry Mumber 1308-04-9 includes 7 digits separated into 3 groups by hyphens. The first part of the number,starting from the left, has 4 digits, 1,3,0 and 8 respectively; the second part has 2 digits, 0 and 4 respectively.
Calculate Digit Verification of CAS Registry Number 1308-04:
(6*1)+(5*3)+(4*0)+(3*8)+(2*0)+(1*4)=49
49 % 10 = 9
So 1308-04-9 is a valid CAS Registry Number.