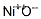1314-06-3 Usage
Uses
Used in Storage Batteries:
Nickel oxide is used as an electrode in fuel cells, specifically in nickel-iron batteries, where it serves as the active material of the negative electrode. It is also utilized in Ni-Cd rechargeable batteries and Ni-MH batteries, playing a crucial role in their functionality.
Used in Chemical Manufacture:
In the chemical industry, nickel oxide is used as a source of nickel for the production of nickel salts and other nickel compounds. It is also employed in the manufacture of metallic nickel and nickel-steel alloys.
Used in Ceramic Industry:
Nickel oxide is used in the ceramic industry for making frit, ferrites, and coloring porcelain. The oxide in sinter form is used in the production of nickel-steel alloys. It supplies oxygen to the melt for the removal of carbon as carbon dioxide.
Used in Ceramic Pigments:
Nickel oxide is used for coloring ceramics and glass, providing a bluish-violet tint in potash glasses and a violet-brown tint in soda glasses. It is considered a powerful colorant, as even a small amount can produce a recognizable tint. Nickel oxide is also used to decolorize potash glass, offering an advantage over manganese dioxide due to its reduced sensitivity to changes in oxidizing and reducing environments.
Used in Tandem Solar Cells:
In the field of solar energy, nickel oxide is used as a photocathode material in tandem solar cells, enhancing their efficiency.
Used in Electronic Devices:
Nickel oxide is employed in electrochromic devices as an anodic electrochromic material, which allows for the control of light transmission through the device.
Used as a Catalyst:
Nickel oxide is also used as a catalyst in hydrogenation reactions, contributing to various chemical processes.
Hazard
Confirmed carcinogen.
Safety Profile
Confirmed human
carcinogen. Poison by subcutaneous route.
Mutation data reported. Hazardous reaction
with hydrogen peroxide. Presence of the
oxide increases the sensitivity of
nitroalkanes (e.g., nitromethane, nitroethane,
1 -nitropropane) to heat. See also NICKEL
COMPOUNDS and PEROXIDES.
Check Digit Verification of cas no
The CAS Registry Mumber 1314-06-3 includes 7 digits separated into 3 groups by hyphens. The first part of the number,starting from the left, has 4 digits, 1,3,1 and 4 respectively; the second part has 2 digits, 0 and 6 respectively.
Calculate Digit Verification of CAS Registry Number 1314-06:
(6*1)+(5*3)+(4*1)+(3*4)+(2*0)+(1*6)=43
43 % 10 = 3
So 1314-06-3 is a valid CAS Registry Number.