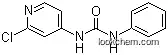68157-60-8 Usage
Uses
Used in Plant Growth Regulation:
Forchlorfenuron is used as a plant growth regulator (PGR) in horticulture. It is commonly used to stimulate the growth of kiwi fruit and grapes, enhancing their yield and quality.
Used in Scientific Research:
Forchlorfenuron is utilized as a research tool to investigate the physiological role of septin and its complexes, providing valuable insights into the mechanisms of cell division and migration.
Trade name
CN-11-3183; KT-30?; SKW 20010
Potential Exposure
Phenylurea/substituted urea plant
growth regulator widely used in agriculture on fruits to
increase their size, to promote cell division, and to improve
the quality and the yield of fruits, especially table grapes,
grape raisins, and kiwi fruit. In some parts of California,
forchlorfenuron is reputed to double the size of Thompson
seedless grapes and delay crop maturity up to several
weeks.
Incompatibilities
May react with strong oxidizers such as
chlorates, peroxides, nitrates, etc. Dust may form explosive
mixture with air.
Waste Disposal
Containers must be disposed
of properly by following package label directions or by
contacting your local or federal environmental control
agency, or by contacting your regional EPA office. If this
material cannot be disposed of according to label instruc tions, it may be dissolved or mixed with a combustible sol vent and burned in a chemical incinerator equipped with an
afterburner and scrubber. In accordance with 40CFR165,
follow recommendations for the disposal of pesticides and
pesticide containers.
References
1) Hu et al. (2008), Forchlorfenuron alters mammalian septin assembly, organization, and dynamics; J. Biol. Chem., 283 29563
2) DeMay et al. (2010), Cellular requirements for the small molecule forchlorfenuron to stabilize the septin cytoskeleton; Cytoskeleton, 67 383
3) Wasik et al. (2012), Septin 7 forms a complex with CD2AP and nephrin and regulates glucose transporter trafficking; Mol. Biol. Cell, 23 3370
4) Vardi-Oknin et al. (2013), Forchlorfenuron disrupts SEPT9_i1 filaments and inhibits HIF-1; PLoS One, 8(8) e73179
Check Digit Verification of cas no
The CAS Registry Mumber 68157-60-8 includes 8 digits separated into 3 groups by hyphens. The first part of the number,starting from the left, has 5 digits, 6,8,1,5 and 7 respectively; the second part has 2 digits, 6 and 0 respectively.
Calculate Digit Verification of CAS Registry Number 68157-60:
(7*6)+(6*8)+(5*1)+(4*5)+(3*7)+(2*6)+(1*0)=148
148 % 10 = 8
So 68157-60-8 is a valid CAS Registry Number.
68157-60-8Relevant academic research and scientific papers
Regioselective preparation of pyridin-2-yl ureas from 2-chloropyridines catalyzed by Pd(0)
Abad, Antonio,Agullo, Consuelo,Cunat, Ana Carmen,Vilanova, Cristina
, p. 915 - 924 (2007/10/03)
The palladium-catalyzed ureidation reaction of 2-chloropyridines can be regioselectively performed in good yield, with both aryl and aliphatic ureas, using xantphos as the ligand, Pd(OAc)2 as the source of palladium, NaOt-Bu/H2O or N
Patch preparations for treating plants
-
, (2008/06/13)
The following invention introduces a patch preparation for treating plants, whereas the patch preparations comprise a chemical layer composed of at least one agrochemically active compound, at least one adhesive and optionally, one or more additives. The components are dispersed in a matrix state on a substrate which are then introduced on the roots of the plant to be treated.