Related Products
- 5-FLUOROTRYPTAMINE
- 5-Fluorotryptamine hydrochloride
- 57618-26-5Calcium bis((S)-3-phenyllactate)
- 576-19-2Biocytin
- 576-22-7Benzene,2-bromo-1,3-dimethyl-
- 576-23-82,3-Dimethylbromobenzene
- 576-24-9Phenol, 2,3-dichloro-
- 57-62-52-Naphthacenecarboxamide,7-chloro-4-(dimethylamino)-1,4,4a,5,5a,6,11,12a-octahydro-3,6,10,12,12a-pentahydroxy-6-methyl-1,11-dioxo-,(4S,4aS,5aS,6S,12aS)-
- 5762-56-1Methanetriamine,N,N,N',N',N'',N''-hexamethyl-
- 57625-74-8Benzeneacetic acid, a,a-dimethyl-, methyl ester
- 57625-97-52-(2,2-DIPHENYLCYCLOPROPYL)-4,5-DIHYDRO-1H-IMIDAZOLE SUCCINATE (2:1)
- 576-26-1Phenol,2,6-dimethyl-
Hot Products
- 104987-11-3Tacrolimus
- 141-53-7Sodium formate
- 8001-54-5Quaternary ammonium compounds, alkylbenzyldimethyl, chlorides
- 9003-39-8Povidone
- 10161-34-9Trenbolone acetate
- 402957-28-2Telaprevir
- 68-19-9Cyanocobalamin
- 7631-86-9Silicon dioxide
- 302-79-4Tretinoin
- 77-92-9Citric acid

|
Basic Information |

|
Post buying leads |

|
Suppliers |

|
Cas Database |

| Name |
5-FLUOROTRYPTAMINE |
EINECS | N/A |
| CAS No. | 576-16-9 | Density | 1.25 g/cm3 |
| PSA | 41.81000 | LogP | 2.50850 |
| Solubility | N/A | Melting Point |
273-275 °C |
| Formula | C10H11FN2 | Boiling Point | 345.959 °C at 760 mmHg |
| Molecular Weight | 178.209 | Flash Point | 163.03 °C |
| Transport Information | N/A | Appearance | N/A |
| Safety | 26-36 | Risk Codes | 20/21/22-36/37/38 |
| Molecular Structure |
|
Hazard Symbols |
 Xn, Xn,  Xi Xi
|
| Synonyms |
Indole,3-(2-aminoethyl)-5-fluoro- (6CI,7CI,8CI);2-(5-Fluoro-1H-indol-3-yl)ethylamine;2-(5-Fluoroindol-3-yl)ethylamine; |
Article Data | 21 |
5-FLUOROTRYPTAMINE Synthetic route
- 208645-53-8
5-fluoro-3-(2-nitrovinyl)indole

- 576-16-9
5-fluorotryptamine
Conditions

| Conditions | Yield |
|---|---|
| With lithium aluminium tetrahydride In tetrahydrofuran at 0 - 20℃; for 40h; | 92% |
| With lithium aluminium tetrahydride In tetrahydrofuran Reflux; |
- 214417-26-2
(Ε)-5-fluoro-3-(2-nitroethenyl)-1Η-indole

- 576-16-9
5-fluorotryptamine
Conditions

| Conditions | Yield |
|---|---|
| With lithium aluminium tetrahydride In tetrahydrofuran at 0 - 20℃; Inert atmosphere; | 76% |
| With lithium aluminium tetrahydride In tetrahydrofuran for 5h; Ambient temperature; | |
| With lithium aluminium tetrahydride In tetrahydrofuran at 20℃; for 5h; |
Conditions

| Conditions | Yield |
|---|---|
| With pyridoxal 5'-phosphate; aromatic L-amino acid decarboxylase In various solvent(s) at 30℃; for 48h; | 41% |
| With NH4OH-NH4Cl buffer; pyridoxal 5'-phosphate at 30℃; for 0.5h; relative rate of CO2 evolution by aromatic L-amino acid decarboxylase from Micrococcus percitreus; |

- 576-16-9
5-fluorotryptamine
Conditions

| Conditions | Yield |
|---|---|
| Stage #1: 5-fluoro-3-(2-nitroethyl)-1Η-indole With ammonium chloride In tetrahydrofuran; ethanol at 85℃; for 0.166667h; Stage #2: With iron | 26.2% |
Conditions

| Conditions | Yield |
|---|---|
| With hydrogenchloride |
- 6346-09-4
4-aminobutyrylaldehyde diethylacetal

- 371-14-2
4-fluorophenylhydrazine

- 576-16-9
5-fluorotryptamine
Conditions

| Conditions | Yield |
|---|---|
| With zinc(II) chloride at 180℃; |
Conditions

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1.1: POCl3 / 0.25 h / 20 °C 1.2: CaCO3 / 1,2-dichloro-ethane / 0.5 h / Heating 2.1: 60 percent / ammonium acetate / 1.5 h / Heating 3.1: LiAlH4 / tetrahydrofuran / 5 h / 20 °C View Scheme | |
| Multi-step reaction with 3 steps 1: 1.) POCl3, 2.) CaCO3 / 1.) RT, 15 min, 2.) 1,2-dichloroethane, reflux, 30 min 2: 60 percent / ammonium acetate / 1.5 h / Heating 3: LiAlH4 / tetrahydrofuran / 5 h / Ambient temperature View Scheme | |
| Multi-step reaction with 3 steps 1.1: trichlorophosphate / 1.25 h / 0 - 38 °C 1.2: 0.08 h / 170 °C 2.1: ammonium acetate / 1.5 h / Reflux 3.1: lithium aluminium tetrahydride / tetrahydrofuran / 40 h / 0 - 20 °C View Scheme | |
| Multi-step reaction with 4 steps 1: diethyl ether / 3 h / 0 - 20 °C 2: diethyl ether / 1 h / 20 °C 3: ammonia / water / 24 h / 20 °C 4: lithium aluminium tetrahydride / tetrahydrofuran / 10 h / Reflux View Scheme |
Conditions

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: 60 percent / ammonium acetate / 1.5 h / Heating 2: LiAlH4 / tetrahydrofuran / 5 h / 20 °C View Scheme | |
| Multi-step reaction with 2 steps 1: 60 percent / ammonium acetate / 1.5 h / Heating 2: LiAlH4 / tetrahydrofuran / 5 h / Ambient temperature View Scheme | |
| Multi-step reaction with 2 steps 1: piperidine; acetic acid / benzene / Reflux 2: lithium aluminium tetrahydride / tetrahydrofuran / Reflux View Scheme | |
| Multi-step reaction with 2 steps 1: ammonium acetate / 1.5 h / Reflux 2: lithium aluminium tetrahydride / tetrahydrofuran / 40 h / 0 - 20 °C View Scheme |
Conditions

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: aqueous HCl; acetic acid 2: aq.-ethanolic KOH 3: aqueous HCl View Scheme |
Conditions

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: aq.-ethanolic KOH 2: aqueous HCl View Scheme |
5-FLUOROTRYPTAMINE Chemical Properties
Product Name: 5-Fluorotryptamine
CAS: 576-16-9
Formula: C10H11FN2
Structure of 5-Fluorotryptamine(576-16-9 ):
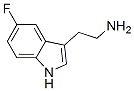
Molecular weight: 178.21
Boiling point: 346 °C at 760 mmHg
Density: 1.249 g/cm3
Flash point: 163 °C
Index of refraction: 1.643
Molar refractivity: 51.6 cm3
Molar volume: 142.5 cm3
Polarizability: 20.45 10-24cm3
Surface tension: 52.3 dyne/cm
Enthalpy of vaporization: 59.01 kJ/mol
Vapour pressure: 5.95E-05 mmHg at 25°C
Water solubility: 3.2e+004(mg/L) at 25 °C
The chemical synonyms of 5-Fluorotryptamine( 576-16-9 ) are 5-FLUOROTRYPTAMINE;AKOS B030092;3-(2-AMINOETHYL)-5-FLUORO-1H-INDOLE;2-(5-FLUORO-1H-INDOL-3-YL)-ETHYLAMINE;RARECHEM AN KA 1417;3-(2-Aminoethyl)-5-fluoro-1H-indole hydrochloride;5-FLUOROTRYPTAMINE,HPLC 98%.
The classification of 5-Fluorotryptamine ( 576-16-9 ) are: Indole/indoline/oxindole;Indole and Indoline;Indoline & Oxindole;Indole.
CAS: 576-16-9
Formula: C10H11FN2
Structure of 5-Fluorotryptamine(576-16-9 ):

Molecular weight: 178.21
Boiling point: 346 °C at 760 mmHg
Density: 1.249 g/cm3
Flash point: 163 °C
Index of refraction: 1.643
Molar refractivity: 51.6 cm3
Molar volume: 142.5 cm3
Polarizability: 20.45 10-24cm3
Surface tension: 52.3 dyne/cm
Enthalpy of vaporization: 59.01 kJ/mol
Vapour pressure: 5.95E-05 mmHg at 25°C
Water solubility: 3.2e+004(mg/L) at 25 °C
The chemical synonyms of 5-Fluorotryptamine( 576-16-9 ) are 5-FLUOROTRYPTAMINE;AKOS B030092;3-(2-AMINOETHYL)-5-FLUORO-1H-INDOLE;2-(5-FLUORO-1H-INDOL-3-YL)-ETHYLAMINE;RARECHEM AN KA 1417;3-(2-Aminoethyl)-5-fluoro-1H-indole hydrochloride;5-FLUOROTRYPTAMINE,HPLC 98%.
The classification of 5-Fluorotryptamine ( 576-16-9 ) are: Indole/indoline/oxindole;Indole and Indoline;Indoline & Oxindole;Indole.
5-FLUOROTRYPTAMINE Safety Profile
Safety information about 5-Fluorotryptamine ( 576-16-9 ):
Hazard Codes
Xi:
Xn:
Risk statements about 5-Fluorotryptamine .
R20/21/22: Harmful by inhalation, in contact with skin and if swallowed .
R36/37/38: Irritating to eyes, respiratory system and skin .
Safety statements about 5-Fluorotryptamine .
S26: In case of contact with eyes, rinse immediately with plenty of water and seek medical advice.
S36: Wear suitable protective clot.
Hazard Codes
Xi:
Xn:

Risk statements about 5-Fluorotryptamine .
R20/21/22: Harmful by inhalation, in contact with skin and if swallowed .
R36/37/38: Irritating to eyes, respiratory system and skin .
Safety statements about 5-Fluorotryptamine .
S26: In case of contact with eyes, rinse immediately with plenty of water and seek medical advice.
S36: Wear suitable protective clot.

