
|
Basic Information |

|
Post buying leads |

|
Suppliers |

|
Cas Database |
- Name Oxindole
- EINECS200-429-5
- CAS No. 59-48-3
- Density1.198 g/cm3
- PSA29.10000
- LogP1.31920
- Solubilityinsoluble in water
- Melting Point123-128 °C(lit.)
- FormulaC8H7NO
- Boiling Point312.7 °C at 760 mmHg
- Molecular Weight133.15
- Flash Point177.8 °C
- Transport InformationUN 2811
- Appearancebeige to yellowish-orange or pinkish-brown powder
- Safety22-24/25
- Risk Codes22
- Molecular Structure
- Hazard Symbols
 Xn
Xn - Synonyms
 Xn
Xn - Article Data216
Oxindole Synthetic route

| Conditions | Yield |
|---|---|
| Stage #1: indole-2,3-dione With hydrazine In methanol Heating; Stage #2: With sodium ethanolate In ethanol Wolff-Kishner reduction; Heating; Further stages.; | 98% |
| With hydrogen In ethanol at 70℃; under 1875.19 Torr; for 0.333333h; | 96.5% |
| With titanium tetrachloride; zinc In tetrahydrofuran at 20℃; for 0.0833333h; Temperature; Reagent/catalyst; Inert atmosphere; | 94% |

| Conditions | Yield |
|---|---|
| With iron; acetic acid at 100℃; | 95% |
| With palladium on activated charcoal; hydrogen; acetic acid at 20℃; for 4h; | 88% |
| With 5%-palladium/activated carbon; hydrogen In ethanol; water under 7500.75 Torr; for 24h; Autoclave; | 88% |
- 30095-98-8
methyl (2-nitrophenyl)acetate

- 59-48-3
2-oxoindole

| Conditions | Yield |
|---|---|
| Stage #1: methyl (2-nitrophenyl)acetate With hydrogen In ethyl acetate at 20℃; under 7600.51 Torr; for 24h; Autoclave; Stage #2: In ethyl acetate for 6h; Reflux; Molecular sieve; | 99% |
| With iron; ammonium chloride; acetic acid at 100℃; for 24h; | 87.8% |
| Multi-step reaction with 2 steps 1: palladium 10% on activated carbon; hydrogen / methanol / 20 °C / Inert atmosphere 2: potassium carbonate / N,N-dimethyl-formamide / 20 °C View Scheme | |
| Multi-step reaction with 2 steps 1: palladium 10% on activated carbon; hydrogen / methanol / 25 °C / Inert atmosphere 2: potassium carbonate / N,N-dimethyl-formamide / 25 °C View Scheme | |
| Multi-step reaction with 2 steps 1: palladium 10% on activated carbon; hydrogen / ethyl acetate / 20 °C / 2280.15 Torr / Sealed tube 2: ethyl acetate / 6 h / Reflux; Molecular sieve View Scheme |

| Conditions | Yield |
|---|---|
| With chloro(meso-tetrakis(2,6-dichlorophenyl)porphyrinato)manganese(III); dihydrogen peroxide In acetonitrile at 20℃; for 1h; | A 44% B 8% |
| With oxygen; titanium(IV) oxide In water for 4h; Irradiation; | |
| With D-glucose; GDH; cytochrome P450 monooxygenase CYP116B4 from Labrenzia aggregata; nicotinamide adenine dinucleotide phosphate In aq. buffer at 30℃; for 5h; pH=8.5; Enzymatic reaction; | |
| With nicotinamide adenine dinucleotide phosphate; cytochrome P450 monooxygenase from Labrenzia aggregata In aq. buffer at 30℃; for 5h; pH=8.5; Enzymatic reaction; enantioselective reaction; |
- 1429481-71-9
1-(toluene-4-sulfonyl)-1,3-dihydro-indol-2-one

- 59-48-3
2-oxoindole

| Conditions | Yield |
|---|---|
| With naphthalene radical anion sodium salt In 1,2-dimethoxyethane at -78 - 18℃; Reagent/catalyst; | 90% |

| Conditions | Yield |
|---|---|
| With chloro[2-nitro-5,10,15,20-tetrakis(2,6-diclorophenyl)porphyrinate]manganese(III); chloro[5,10,15,20-tetrakis(4-dimethy-lamino-2,3,5,6-tetrafluorophenyl)porphyrinate]manganese(III) In acetonitrile at 20℃; for 0.5h; | A 35% B 7% |
| With ammonium persulfate; phosphoric acid In water pH=1.4; Oxidation; | A 26% B 32% |
- 31912-02-4
ethyl 2-nitrophenylacetate

- 59-48-3
2-oxoindole

| Conditions | Yield |
|---|---|
| With acetic acid; zinc In ethanol at 70℃; for 4h; Inert atmosphere; | 93% |
| With acetic acid; zinc In ethanol at 70℃; for 4h; | 66% |

| Conditions | Yield |
|---|---|
| With chloro(meso-tetrakis(2,6-dichlorophenyl)porphyrinato)manganese(III); dihydrogen peroxide In acetonitrile at 20℃; for 0.5h; | A 64% B 7% C 6% |

| Conditions | Yield |
|---|---|
| With chloro(meso-tetrakis(2,6-dichlorophenyl)porphyrinato)manganese(III); chloro[2-nitro-5,10,15,20-tetrakis(2,6-diclorophenyl)porphyrinate]manganese(III) In acetonitrile at 20℃; for 0.5h; | A 50% B 6% C 6% |
- 171359-95-8
N'-(2-aminobenzylidene)-4-methylbenzenesulfonohydrazide

- 13939-06-5, 199620-15-0
molybdenum hexacarbonyl

- 59-48-3
2-oxoindole

| Conditions | Yield |
|---|---|
| With tetraethylammonium bromide; 1,8-diazabicyclo[5.4.0]undec-7-ene In 1,4-dioxane at 20 - 110℃; for 1h; Solvent; Reagent/catalyst; | 83% |
Oxindole Chemical Properties
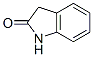
IUPAC Name:1,3-dihydroindol-2-one
Molecular Formula:C8H7NO
Molecular Weight:133.147280 g/mol
Melting Point:123-128 °C(lit.)
Boiling Point:227 °C73 mm Hg(lit.)
Flash Point:194-196°C/17mm
Insolubility:Water
EINECS:200-429-5
BRN:114692
CAS DataBase Reference:59-48-3(CAS DataBase Reference)
NIST Chemistry Reference:59-48-3(NIST)
EPA Substance Registry:System 59-48-3(EPA Substance)
Synonyms of 2-OXINDOLE(59-48-3):
OXINDOL;OXINDOLE;O-(AMINOPHENYL)ACETIC ACID LACTAM;TIMTEC-BB SBB004215;1,3-dihydro-2h-indol-2-on;1,3-Dihydro-indol-2-one;1,3-Dihydroindol-2-one;2,3-dihydroindol-2-one;Oxindole
Categories of 2-OXINDOLE(59-48-3):
blocks;IndolesOxindoles;Aromatic Phenylacetic Acids and Derivatives;Indoles and derivatives;Indoline & Oxindole;Indoles;Indole Derivatives
Oxindole Safety Profile
Hazard Codes:Xn

Risk Statements:22
22:Harmful if swallowed
Safety Statements:22-24/25
22:Do not breathe dust
24/25:Avoid contact with skin and eyes
RIDADR:2811
WGK Germany:3
RTECS:NM2080500
HazardClass:6.1(b)
PackingGroup:III
HS Code:29337900
Oxindole Specification
Keep container closed when not in use. Store in a tightly closed container. Store in a cool, dry, well-ventilated area away from incompatible substances.
First Aid Measures of 2-OXINDOLE(59-48-3):
Ingestion:If victim is conscious and alert, give 2-4 cupfuls of milk or water. Never give anything by mouth to an unconscious person. Get medical aid immediately.
Inhalation:Remove from exposure to fresh air immediately. If not breathing, give artificial respiration. If breathing is difficult, give oxygen. Get medical aid.
Skin:Get medical aid. Flush skin with plenty of soap and water for at least 15 minutes while removing contaminated clothing and shoes. Wash clothing before reuse.
Eyes:Flush eyes with plenty of water for at least 15 minutes, occasionally lifting the upper and lower eyelids. Get medical aid immediately.
- 59483-54-4Benzenamine,3-chloro-2-nitro-
- 59483-84-0Bis(pentafluorophenyl) carbonate
- 59484-60-52-Pyrazinecarboximidicacid, 6-methoxy-, methyl ester
- 59484-69-42-Pyrazinecarboxylic acid, 6-propoxy-
- 594859-54-8Y-39983
- 59486-73-6D-Glutamic acid,N-[(phenylmethoxy)carbonyl]-, 5-(phenylmethyl) ester
- 59488-34-5Hydrazine,(4-bromo-2-nitrophenyl)-
- 59488-60-7Thieno[2,3-β]pyridine-2-carboxylicacid, 3-amino-6-methyl-
- 59489-31-5Quinoxaline,2,7-dichloro-
- 59489-71-32-Amino-5-bromopyrazine
Hot Products
- 104987-11-3Tacrolimus
- 141-53-7Sodium formate
- 8001-54-5Quaternary ammonium compounds, alkylbenzyldimethyl, chlorides
- 9003-39-8Povidone
- 10161-34-9Trenbolone acetate
- 402957-28-2Telaprevir
- 68-19-9Cyanocobalamin
- 7631-86-9Silicon dioxide
- 302-79-4Tretinoin
- 77-92-9Citric acid

