Products Categories
| CAS No.: | 146142-94-1 |
|---|---|
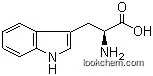
| Name: | Monatin |
| Article Data: | 12 |
| Molecular Structure: | |
|
|
|
| Formula: | C14H16N2O5 |
| Molecular Weight: | 292.291 |
| Synonyms: | (2S,4S)-Monatin;L-Glutamicacid, 4-hydroxy-4-(1H-indol-3-ylmethyl)-, (S)-;(2S,4S)-2-Amino-4-carboxy-4-hydroxy-5-(3-indolyl)pentanoic acid;L-Glutamic acid,4-hydroxy-4-(1H-indol-3-ylmethyl)-, (4S)-; |
| Density: | 1.526 g/cm3 |
| Boiling Point: | 656.358 °C at 760 mmHg |
| Flash Point: | 350.753 °C |
| PSA: | 136.64000 |
| LogP: | 1.02840 |
- 483366-12-7(2S,4R)-1-Boc-2-cyano-4-hydroxypyrrolidine
- 173606-50-3BOC-10-AMINODECANOIC ACID
- 361456-36-2METHYL (R)-(+)-ISOCYANATO-3-PHENYLPROPI&
- 5156-58-1N-(1-Benzyl-4-pipperidinyl)-N-phenylpropanamide HCl
- 81281-59-67-Benzylideneaminotheophylline
- 50288-62-5threo-Phenyl-2-piperidyl acetamide
- 82993-81-5D-threo-Ritalinic acid hydrochloride
- 47087-37-6Z-D-Glu-OMe
- 73441-42-6METHYL-5-CHLORO-2,2-DIMETHYLVALERATE
- 211914-50-0N-[[2-[[[4-(Aminoiminomethyl)phenyl]amino]methyl]-1-methyl-1H-benzimidazol-5-yl]carbonyl]-N-(2-pyridinyl)-beta-alanine ethyl ester hydrochloride

- 146142-94-1
(2S,4S)-2-((1H-indol-3-yl)methyl)-4-amino-2-hydroxypentane-1,5-dioic acid

| Conditions | Yield |
|---|---|
| With sodium hydroxide In methanol | 92% |
| With sodium hydroxide In methanol; water at 20℃; for 6h; | 54 mg |
| Stage #1: (2S,4S)-4-Amino-2-(1H-indol-3-ylmethyl)-5-oxo-tetrahydro-furan-2-carboxylic acid With water; potassium hydroxide In methanol at 25℃; for 1h; Inert atmosphere; Large scale reaction; Stage #2: With acetic acid In methanol pH=6.5; Large scale reaction; Stage #3: With acetic acid In methanol pH=6.5; Large scale reaction; optical yield given as %ee; | 193 g |
- 855433-72-6
4-amino-2-hydroxy-2-(1H-indol-3-ylmethyl)-pentanedioic acid

A
- 400769-77-9
(2S,4R) 4-hydroxy-4-(3-indolylmethyl)-2-aminoglutaric acid

B
- 146142-94-1
(2S,4S)-2-((1H-indol-3-yl)methyl)-4-amino-2-hydroxypentane-1,5-dioic acid

| Conditions | Yield |
|---|---|
| RP18-HPLC resolution; |
- 551958-84-0
4-hydroxy-4-(3-indolylmethyl)-2-ketoglutaric acid

- 146142-94-1
(2S,4S)-2-((1H-indol-3-yl)methyl)-4-amino-2-hydroxypentane-1,5-dioic acid

| Conditions | Yield |
|---|---|
| With pyridoxal 5'-phosphate; tris hydrochloride; monosodium glutamate; Aeromonas hydrophila IFO3820 In water; toluene at 30℃; for 16h; pH=7.6; Product distribution / selectivity; Enzymatic reaction; Culture medium; | |
| With pyridoxal 5'-phosphate; tris hydrochloride; monosodium glutamate; Agrobacterium tumefaciens IFO3058 In water; toluene at 30℃; for 16h; pH=7.6; Product distribution / selectivity; Enzymatic reaction; Culture medium; | |
| With pyridoxal 5'-phosphate; tris hydrochloride; monosodium glutamate; Alcaligenes faecalis ATCC8750 In water; toluene at 30℃; for 16h; pH=7.6; Product distribution / selectivity; Enzymatic reaction; Culture medium; |
- 73-22-3
L-Tryptophan

- 146142-94-1
(2S,4S)-2-((1H-indol-3-yl)methyl)-4-amino-2-hydroxypentane-1,5-dioic acid

| Conditions | Yield |
|---|---|
| With potassium phosphate; pyridoxal 5'-phosphate; magnesium chloride; Comamonas testosteroni proA Aldolase; HIS6-HEXaspC aminotransferase In water for 27 - 29h; pH=7.8; Product distribution / selectivity; Enzymatic reaction; | |
| With potassium phosphate; pyridoxal 5'-phosphate; magnesium chloride; Comamonas testosteroni proA Aldolase; HIS6-HEXaspC aminotransferase In water for 27 - 29h; pH=7.8; Product distribution / selectivity; Enzymatic reaction; |
- 73-22-3
L-Tryptophan

A
- 914081-89-3
(S)-2-hydroxy-2-(1H-indol-3-ylmethyl)-4-oxo-pentanedioic acid

B
- 146142-94-1
(2S,4S)-2-((1H-indol-3-yl)methyl)-4-amino-2-hydroxypentane-1,5-dioic acid

| Conditions | Yield |
|---|---|
| With potassium phosphate; pyridoxal 5'-phosphate; magnesium chloride; Comamonas testosteroni proA Aldolase; HIS6-HEXaspC aminotransferase In water at 30℃; for 3 - 5h; pH=7.5 - 7.8; Product distribution / selectivity; Enzymatic reaction; |
- 56-41-7
L-alanin

A
- 914081-89-3
(S)-2-hydroxy-2-(1H-indol-3-ylmethyl)-4-oxo-pentanedioic acid

B
- 146142-94-1
(2S,4S)-2-((1H-indol-3-yl)methyl)-4-amino-2-hydroxypentane-1,5-dioic acid

| Conditions | Yield |
|---|---|
| Stage #1: L-Tryptophan With potassium phosphate; pyridoxal 5'-phosphate; magnesium chloride; Comamonas testosteroni proA Aldolase; HIS6-HEXaspC aminotransferase In water at 30℃; for 3 - 5h; pH=7.5 - 7.8; Enzymatic reaction; Stage #2: L-alanin; HIS6-HEXaspC aminotransferase In water at 30℃; pH=7.5 - 7.8; Product distribution / selectivity; Enzymatic reaction; |
- 113-24-6
sodium pyruvate

- 73-22-3
L-Tryptophan

- 146142-94-1
(2S,4S)-2-((1H-indol-3-yl)methyl)-4-amino-2-hydroxypentane-1,5-dioic acid

| Conditions | Yield |
|---|---|
| With potassium phosphate; pyridoxal 5'-phosphate; magnesium chloride; Comamonas testosteroni proA Aldolase; HIS6-HEXaspC aminotransferase In water at 30℃; for 21 - 29h; pH=7.8; Product distribution / selectivity; Enzymatic reaction; |

- 146142-94-1
(2S,4S)-2-((1H-indol-3-yl)methyl)-4-amino-2-hydroxypentane-1,5-dioic acid

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1: 15.6 mg / NaHCO3 / acetonitrile; H2O / 24 h / 20 °C 2: 69 percent / PDC / dimethylformamide / 24 h / 20 °C 3: HCO2H; HCl / 4 h / 20 °C 4: 54 mg / NaOH / methanol; H2O / 6 h / 20 °C View Scheme |
- 645396-51-6
2-[(indol-3-yl)methyl]prop-2-en-1-ol

- 146142-94-1
(2S,4S)-2-((1H-indol-3-yl)methyl)-4-amino-2-hydroxypentane-1,5-dioic acid

| Conditions | Yield |
|---|---|
| Multi-step reaction with 9 steps 1: CH2Cl2 / 360 h / 20 °C 2: 97 percent / imidaziole / dimethylformamide / 3 h / 20 °C 3: 97 percent / DMAP / acetonitrile / 1 h / 20 °C 4: 100 percent / pyridine*HF / tetrahydrofuran / 1 h / 0 °C 5: H2 / Pd(OH)2/C / methanol / 5 h / 20 °C / atmospheric pressure 6: 15.6 mg / NaHCO3 / acetonitrile; H2O / 24 h / 20 °C 7: 69 percent / PDC / dimethylformamide / 24 h / 20 °C 8: HCO2H; HCl / 4 h / 20 °C 9: 54 mg / NaOH / methanol; H2O / 6 h / 20 °C View Scheme | |
| Multi-step reaction with 9 steps 1: 98 percent / MgBr2*OEt2 / CH2Cl2 / 84 h / 20 °C 2: 97 percent / imidaziole / dimethylformamide / 3 h / 20 °C 3: 97 percent / DMAP / acetonitrile / 1 h / 20 °C 4: 100 percent / pyridine*HF / tetrahydrofuran / 1 h / 0 °C 5: H2 / Pd(OH)2/C / methanol / 5 h / 20 °C / atmospheric pressure 6: 15.6 mg / NaHCO3 / acetonitrile; H2O / 24 h / 20 °C 7: 69 percent / PDC / dimethylformamide / 24 h / 20 °C 8: HCO2H; HCl / 4 h / 20 °C 9: 54 mg / NaOH / methanol; H2O / 6 h / 20 °C View Scheme | |
| Multi-step reaction with 9 steps 1: CH2Cl2 / 360 h / 20 °C 2: 97 percent / imidazole / dimethylformamide 3: 97 percent / DMAP / acetonitrile 4: 100 percent / HF*pyridine / tetrahydrofuran 5: H2 / Pd(OH)2/C / methanol 6: acetonitrile 7: 69 percent / PDC / dimethylformamide 8: HCl; HCOOH 9: 92 percent / NaOH / methanol View Scheme | |
| Multi-step reaction with 9 steps 1: 98 percent / MgBr2*OEt2 / CH2Cl2 / 72 h / 20 °C 2: 97 percent / imidazole / dimethylformamide 3: 97 percent / DMAP / acetonitrile 4: 100 percent / HF*pyridine / tetrahydrofuran 5: H2 / Pd(OH)2/C / methanol 6: acetonitrile 7: 69 percent / PDC / dimethylformamide 8: HCl; HCOOH 9: 92 percent / NaOH / methanol View Scheme |
- 87438-91-3
ethyl 2-((1H-indol-3-yl)methyl)acrylate

- 146142-94-1
(2S,4S)-2-((1H-indol-3-yl)methyl)-4-amino-2-hydroxypentane-1,5-dioic acid

| Conditions | Yield |
|---|---|
| Multi-step reaction with 10 steps 1: 85 percent / DIBAL-H / CH2Cl2; hexane / -78 - 20 °C 2: CH2Cl2 / 360 h / 20 °C 3: 97 percent / imidaziole / dimethylformamide / 3 h / 20 °C 4: 97 percent / DMAP / acetonitrile / 1 h / 20 °C 5: 100 percent / pyridine*HF / tetrahydrofuran / 1 h / 0 °C 6: H2 / Pd(OH)2/C / methanol / 5 h / 20 °C / atmospheric pressure 7: 15.6 mg / NaHCO3 / acetonitrile; H2O / 24 h / 20 °C 8: 69 percent / PDC / dimethylformamide / 24 h / 20 °C 9: HCO2H; HCl / 4 h / 20 °C 10: 54 mg / NaOH / methanol; H2O / 6 h / 20 °C View Scheme | |
| Multi-step reaction with 10 steps 1: 85 percent / DIBAL-H / CH2Cl2; hexane / -78 - 20 °C 2: 98 percent / MgBr2*OEt2 / CH2Cl2 / 84 h / 20 °C 3: 97 percent / imidaziole / dimethylformamide / 3 h / 20 °C 4: 97 percent / DMAP / acetonitrile / 1 h / 20 °C 5: 100 percent / pyridine*HF / tetrahydrofuran / 1 h / 0 °C 6: H2 / Pd(OH)2/C / methanol / 5 h / 20 °C / atmospheric pressure 7: 15.6 mg / NaHCO3 / acetonitrile; H2O / 24 h / 20 °C 8: 69 percent / PDC / dimethylformamide / 24 h / 20 °C 9: HCO2H; HCl / 4 h / 20 °C 10: 54 mg / NaOH / methanol; H2O / 6 h / 20 °C View Scheme | |
| Multi-step reaction with 11 steps 1: 97 percent / Et3N; 4-DMAP / CH2Cl2 / 20 °C 2: 72 percent / DIBAL-H / CH2Cl2 / -78 °C 3: MgBr2*OEt2 / CH2Cl2 / 96 h / 20 °C 4: 97 percent / imidazole / dimethylformamide 5: 97 percent / DMAP / acetonitrile 6: 100 percent / HF*pyridine / tetrahydrofuran 7: H2 / Pd(OH)2/C / methanol 8: acetonitrile 9: 69 percent / PDC / dimethylformamide 10: HCl; HCOOH 11: 92 percent / NaOH / methanol View Scheme |
- Total:20 Page 1 of 1 1
Specification
The Monatin, with the CAS registry number 146142-94-1, is also known as L-Glutamic acid,4-hydroxy-4-(1H-indol-3-ylmethyl)-, (4S)-. This chemical's molecular formula is C14H16N2O5 and molecular weight is 292.29. What's more, its IUPAC name is (2S,4S)-4-amino-2-hydroxy-2-(1H-indol-3-ylmethyl)pentanedioic acid. It is a naturally-occurring, high intensity sweetener isolated from the plant sclerochiton ilicifolius. It contains no carbohydrate or sugar, and nearly no food energy, unlike sucrose or other nutritive sweeteners.
Physical properties of Monatin are: (1)ACD/LogP: 0.90; (2)# of Rule of 5 Violations: 1; (3)ACD/LogD (pH 5.5): -3; (4)ACD/LogD (pH 7.4): -3; (5)ACD/BCF (pH 5.5): 1; (6)ACD/BCF (pH 7.4): 1; (7)ACD/KOC (pH 5.5): 1; (8)ACD/KOC (pH 7.4): 1; (9)#H bond acceptors: 7; (10)#H bond donors: 6; (11)#Freely Rotating Bonds: 8; (12)Polar Surface Area: 136.64 Å2; (13)Index of Refraction: 1.708; (14)Molar Refractivity: 74.733 cm3; (15)Molar Volume: 191.575 cm3; (16)Polarizability: 29.626×10-24cm3; (17)Surface Tension: 88.913 dyne/cm; (18)Density: 1.526 g/cm3; (19)Flash Point: 350.753 °C; (20)Enthalpy of Vaporization: 101.55 kJ/mol; (21)Boiling Point: 656.358 °C at 760 mmHg.
You can still convert the following datas into molecular structure:
(1)SMILES: O=C(O)[C@@H](N)C[C@](O)(C(=O)O)Cc2c1ccccc1nc2
(2)InChI: InChI=1S/C14H16N2O5/c15-10(12(17)18)6-14(21,13(19)20)5-8-7-16-11-4-2-1-3-9(8)11/h1-4,7,10,16,21H,5-6,15H2,(H,17,18)(H,19,20)/t10-,14-/m0/s1
(3)InChIKey: RMLYXMMBIZLGAQ-HZMBPMFUSA-N