Products Categories
| CAS No.: | 552-89-6 |
|---|---|
| Name: | 2-Nitrobenzaldehyde |
| Article Data: | 462 |
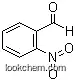
| Molecular Structure: | |
|
|
|
| Formula: | C7H5NO3 |
| Molecular Weight: | 151.122 |
| Synonyms: | Benzaldehyde, o-nitro-;Benzaldehyde, 2-nitro-;o-Nitrobenzaldehyde;4-chlorine-3- nitro acetophenone;O-Nitro Benzaldehyde;o-Nitrobenzyldehyde; |
| EINECS: | 209-025-3 |
| Density: | 1.338 g/cm3 |
| Melting Point: | 42-44 °C(lit.) |
| Boiling Point: | 268.2 °C at 760 mmHg |
| Flash Point: | 144 °C |
| Solubility: | Insoluble in water. |
| Appearance: | Yellow crystalline powder or needles |
| Hazard Symbols: |
 Xn, Xn,  Xi Xi
|
| Risk Codes: | 22-36/37/38-68 |
| Safety: | 26-24/25-36/37/39-36 |
| PSA: | 62.89000 |
| LogP: | 1.93050 |
- 81281-59-67-Benzylideneaminotheophylline
- 82993-81-5D-threo-Ritalinic acid hydrochloride
- 73441-42-6METHYL-5-CHLORO-2,2-DIMETHYLVALERATE
- 68439-39-4Poly(oxy-1,2-ethanediyl), alpha-(2-ethylhexyl)-omega-hydroxy-,
- 852475-26-4MC1568
- 958254-66-51H-Imidazo[4,5-b]pyridine-2-carboxaldehyde, 1-methyl-, hydrochloride
- 99170-93-1N-Methyl-2-oxazolamine
- 717878-06-31-(4-fluorophenyl)-4-nitro-1H-imidazole
- 914458-26-7[5-(2-fluorophenyl)-1-pentyl-1H-pyrrol-3-yl]-1-naphthalenyl-Methanone
- 894852-01-87-BROMO-2,2-DIMETHYL-2H-PYRIDO[3,2-B][1,4]OXAZIN-3(4H)-ONE

| Conditions | Yield |
|---|---|
| With [NO(1+)*18-crown-6*H(NO3)2(1-)]; silica gel In dichloromethane at 20℃; for 0.0833333h; | 100% |
| With beta zeolite modified with chlorosulphonic acid (28 wtpercent) In ethanol at 50℃; for 0.5h; | 100% |
| With poly(4-vinylpyridine)-supported sulfuric acid In acetonitrile at 50℃; for 1h; Green chemistry; | 100% |

| Conditions | Yield |
|---|---|
| With oxygen; perruthenate modified mesoporous silicate MCM-41 In toluene at 80℃; for 3h; Oxidation; | 100% |
| With butyltriphenylphosphonium chlorochromate In acetonitrile for 2.5h; Heating; | 100% |
| With 1H-imidazole; [bis(acetoxy)iodo]benzene In dichloromethane at 20℃; for 2h; | 100% |

- 552-89-6
2-nitro-benzaldehyde

| Conditions | Yield |
|---|---|
| With hydrogenchloride for 0.0416667h; Product distribution; Ambient temperature; pH = 4-6, regeneration of aldehyde; | 100% |
- 62673-13-6
o-Nitrobenzyloxytrimethylsilane

- 552-89-6
2-nitro-benzaldehyde

| Conditions | Yield |
|---|---|
| With nitrogen dioxide at 20℃; for 1h; | 100% |
| With potassium permanganate; tetrachlorosilane In acetonitrile at 20℃; for 0.416667h; | 89% |
| With zinc dichromate(VI) at 20℃; | 89% |
- 51673-59-7
1-(2-nitrophenyl)ethane-1,2-diol

- 552-89-6
2-nitro-benzaldehyde

| Conditions | Yield |
|---|---|
| With [bis(acetoxy)iodo]benzene In dichloromethane at 25℃; for 8h; Inert atmosphere; | 99% |
| With tert-butylhypochlorite; lead acetate; dibenzoyl peroxide In toluene at 20℃; for 0.75h; | 90% |
- 77376-01-3
acetic acid 2-nitro-benzyl ester

- 552-89-6
2-nitro-benzaldehyde

| Conditions | Yield |
|---|---|
| With methanol; potassium permanganate In ethyl acetate at 25℃; for 30h; | 99% |
- 35531-58-9
2-(2-nitrophenyl)-1,3-dithiane

- 552-89-6
2-nitro-benzaldehyde

| Conditions | Yield |
|---|---|
| With 1-benzyl-4-aza-1-azoniabicyclo[2.2.2]octane tribromide In methanol; dichloromethane at 20℃; for 0.05h; | 98% |
| With dihydrogen peroxide; iodine; sodium dodecyl-sulfate In water at 20℃; for 4h; Micellar solution; | 98% |
| With 2,4,4,6-Tetrabromo-2,5-cyclohexadien-1-one; dihydrogen peroxide In water; acetonitrile at 20℃; for 4.5h; | 98% |
- 93304-97-3
((2-nitrophenyl)methylene)bis(phenylsulfane)

- 552-89-6
2-nitro-benzaldehyde

| Conditions | Yield |
|---|---|
| With ammonium iodide; dihydrogen peroxide; acetic acid In water at 20℃; for 1.5h; | 98% |
- 16968-19-7
2,2'-dinitrobibenzyl

- 552-89-6
2-nitro-benzaldehyde

| Conditions | Yield |
|---|---|
| With p-benzoquinone at 120℃; for 8h; Concentration; Temperature; | 97.6% |
| With p-benzoquinone at 140℃; for 5h; Temperature; | 91% |

| Conditions | Yield |
|---|---|
| With manganese(IV) oxide In dichloromethane at 20℃; for 18h; | 97% |
| With (CH3)2NCH2NpSiH2Ph at 110 - 140℃; | 50% |
| With carboxylic acid reductase Kinetics; Green chemistry; Enzymatic reaction; | |
| Multi-step reaction with 3 steps 1: thionyl chloride; N,N-dimethyl-formamide / dichloromethane / 20 °C 2: sodium tetrahydroborate / tetrahydrofuran; N,N-dimethyl-formamide / 0 - 20 °C 3: pyridinium chlorochromate / dichloromethane / 20 °C View Scheme |
Consensus Reports
Specification
The 2-Nitrobenzaldehyde , with the CAS register number 552-89-6, is also known to us as benzaldehyde, o-nitro-;benzaldehyde,2-nitro-;o-nitro-benzaldehyd;2-nitrobenzaldehyde;2-formylnitrobenzene;akos bbs-00003153;akos 93864;nitrobenzaldehyde-2;orthonitro benzaldehyde;o-nitrobenzaldehyde;2-nitobenzaldehyde;o-nitrobbenzaldehyde;o-nitrobenzaldehyde(2-nitrobenzaldehyde);2-nitrobenzaldehyde, for the determ. of methyl ketones;2-nitrobenzaldehydegr;o-nitrobenzaldehyde99.5%;2-nitrobenzaldehyde, 98+%;2-nitrobenzaldehyde, 99+%;o-nitrobenzaldehyde (ortho-nitrobenzaldehyde) .
The physical properties of this kind of chemical are as following: (1)ACD/BCF (pH 5.5): 12.37 ; (2)ACD/BCF (pH 7.4): 12.37 ; (3)ACD/KOC (pH 5.5): 210.65 ; (4)ACD/KOC (pH 7.4): 210.65 ; (5)#H bond acceptors: 4 ; (6)#Freely Rotating Bonds: 2 ; (7)Polar Surface Area: 62.89 ; (8)Index of Refraction: 1.617 ; (9)Molar Refractivity: 39.55 cm3 ; (10)Molar Volume: 112.9 cm3 ; (11)Polarizability: 15.67 ×10-24 cm3 ; (12)Surface Tension: 55.1 dyne/cm ; (13)Density: 1.338 g/cm3 ; (14)Flash Point: 144 °C ; (15)Enthalpy of Vaporization: 50.63 kJ/mol ; (16)Boiling Point: 268.2 °C at 760 mmHg ; (17)Vapour Pressure: 0.0078 mmHg at 25°C .
It is a kind of light yellow acicular crystal with the same odour of benzaldehyde, and is soluble in ethonal and aether, benzene while slightly soluble in water. And it is sensitive to air. Being a kind of harmful chemical, it may cause damage to health; and if swallowed, it will be very harmful. Besides, it is also irritant to eyes, respiratory system and skin and may cause inflammation to the skin or other mucous membranes. What's more, it will have possible risk of irreversible effects. Therefore, while using this chemical, you need to be cautious. Wear suitable protective clothing, gloves and eye/face protection and void contact with skin and eyes. If in case of contact with eyes, rinse immediately with plenty of water and seek medical advice. Then you could also go to WGK Germany : 2 to get more safety information .
This kind of chemical could be used as the organic synthesis intermediate, such as being used to produce the nifedipine which is the antianginal drug in the phamaceutic industry. Besides, it could also to be used in composing quinoline loops drugs when its nitro be turned into amino. When it comes to its production method, you could get it through the following ways. The first way is to react it with acetic anhydride and then hydrolyze and then you could get it; The second way is to nitrify it and then have the oxidation and finally you could get it. If you need to store it, you should keep it at room temperature.
In addition, you could refer to the following data information to get the molecular structure:
SMILES:N#Cc1cccc([N+]([O-])=O)c1
InChI:InChI=1/C7H4N2O2/c8-5-6-2-1-3-7(4-6)9(10)11/h1-4H
InChIKey:RUSAWEHOGCWOPG-UHFFFAOYAS
Below are the toxicity information of this kind of chemcial:
| Organism | Test Type | Route | Reported Dose (Normalized Dose) | Effect | Source |
|---|---|---|---|---|---|
| mouse | LD50 | oral | 600mg/kg (600mg/kg) | BEHAVIORAL: ALTERED SLEEP TIME (INCLUDING CHANGE IN RIGHTING REFLEX) LUNGS, THORAX, OR RESPIRATION: DYSPNEA GASTROINTESTINAL: CHANGES IN STRUCTURE OR FUNCTION OF SALIVARY GLANDS | Annales des Falsifications et de l'Expertise Chimique. Vol. 76(815), Pg. 65, 1983. |