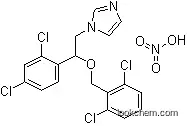
24168-96-5 Usage
Uses
Used in Pharmaceutical Industry:
Isoconazole nitrate is used as an antifungal agent for the treatment of various fungal infections. Its antifungal properties make it effective against a range of fungal pathogens, providing relief and treatment for patients suffering from such conditions.
Used in Local Treatment of Vaginitis:
Isoconazole nitrate is used as an antibacterial in the local treatment of vaginitis. Its antibacterial properties help to eliminate harmful bacteria that cause inflammation and discomfort, promoting a healthier and more balanced vaginal environment.
Originator
Fazol,Fournier,France,1979
Manufacturing Process
To a stirred and refluxing solution of 40 parts of benzene and 35 parts of
dimethylformamide (both solvents previously dried azeotropically) are added
successively 1.6 parts of sodium hydride and 7.7 parts of α-(2,4-
dichlorophenyl)imidazole-1-ethanol, (cooling on ice is necessary). After the
addition is complete, stirring and refluxing is continued for 30 minutes. Then
there are added 7.8 parts of 2.6-dichlorobenzyl chloride and the whole is
stirred at reflux for another 3 hours. The reaction mixture is poured onto
water and the product 1-[2,4-dichloro-b-(2,6-dichlorobenzyloxy)phenethyl]
imidazole, is extracted with benzene. The extract is washed twice with water,
dried, filtered and evaporated in vacuo. The base residue is dissolved in a
mixture of acetone and diisopropyl ether and to this solution is added an
excess of concentrated nitric acid solution. The precipitated nitrate salt is
filtered off and recrystallized from a mixture of methanol and diisopropyl
ether, yielding 1-[2,4-dichloro-b-(2,6-dichlorobenzyloxy)phenethyl]imidazole
nitrate; melting point 179°C.
Check Digit Verification of cas no
The CAS Registry Mumber 24168-96-5 includes 8 digits separated into 3 groups by hyphens. The first part of the number,starting from the left, has 5 digits, 2,4,1,6 and 8 respectively; the second part has 2 digits, 9 and 6 respectively.
Calculate Digit Verification of CAS Registry Number 24168-96:
(7*2)+(6*4)+(5*1)+(4*6)+(3*8)+(2*9)+(1*6)=115
115 % 10 = 5
So 24168-96-5 is a valid CAS Registry Number.
InChI:InChI=1/C18H14Cl4N2O.NO3/c19-12-4-5-13(17(22)8-12)18(9-24-7-6-23-11-24)25-10-14-15(20)2-1-3-16(14)21;2-1(3)4/h1-8,11,18H,9-10H2;/q;-1/p+1
24168-96-5Relevant articles and documents
New copper(II) complexes with isoconazole: Synthesis, structures and biological properties
Dulcevscaia, Galina M.,Kravtsov, Victor Ch.,Macaev, Fliur Z.,Duca, Gheorghe G.,Stingachi, Eugenia P.,Pogrebnoi, Serghei I.,Boldescu, Veaceslav V.,Clapco, Steliana F.,Tiurina, Janeta P.,Deseatnic-Ciloci, Alexandra A.,Lipkowski, Janusz,Liu, Shi-Xia,Decurtins, Silvio,Baca, Svetlana G.
, p. 106 - 114 (2013/06/05)
There is an increasing demand for novel metal-based complexes with biologically relevant molecules in technology and medicine. Three new Cu(II) coordination compounds with antifungal agent isoconazole (L), namely mononuclear complexes [CuCl2(L)2] (1), and [Cu(O2CMe) 2(L)2]·2H2O (2) and coordination polymer [Cu(pht)(L)2]n (3) (where H2pht-o-phthalic acid) were synthesized and characterized by IR spectroscopy, thermogravimetric analysis and X-ray crystallography. X-ray analysis showed that in all complexes, the isoconazole is coordinated to Cu(II) centres by a N atom of the imidazole fragment. In complex 1, the square-planar environment of Cu(II) atoms is completed by two N atoms of isoconazole and two chloride ligands, whereas the Cu(II) atoms are coordinated by two N atoms from two isoconazole ligands and two O atoms from the different carboxylate residues: acetate in 2 and phthalate in 3. The formation of an infinite chain through the bridging phthalate ligand is observed in 3. The biosynthetic ability of micromycetes Aspergillus niger CNMN FD 10 in the presence of the prepared complexes 1-3 as well as the antifungal drug isoconazole were studied. Complexes 2 and 3 accelerate the biosynthesis of enzymes (β-glucosidase, xylanase and endoglucanase) by this fungus. Moreover, a simplified and improved method for the preparation of isoconazole nitrate was developed.